Table of Contents
Introduction:
Soluble base metals have a place with the s-block components involving the furthest left half of the occasional table. Soluble base metals promptly lose electrons, making the most of them among the most receptive components on the planet. In this article, we will make sense of the electronic setups, ionization enthalpy, hydration enthalpy and nuclear, ionic radii and other physical and substance properties of the gathering one salt metals.
What are Alkali Metals?
- Overall ‘antacid’ alludes to the fundamental or soluble nature of their metal hydroxides. The mixtures are called soluble base metals since when they respond with water they normally structure alkalies which are only solid bases that can undoubtedly kill acids.
- Antacid metals have a relating [Noble gas] ns1 electronic design. They possess the main segment of the occasional table. Antacid components are Lithium(Li), Sodium(Na), Potassium (K), Rubidium (Ru), Cesium (Cs) and Francium (Fr) possessing progressive periods from first to seven. Francium is a radioactive component with an exceptionally low half-life.
- Notwithstanding, the fundamental motivation behind why hydrogen (H) isn’t considered as a salt metal is that it is generally found as a gas when the temperature and tension are ordinary. Hydrogen can show properties or change into a salt metal when it is presented to incredibly high tension.
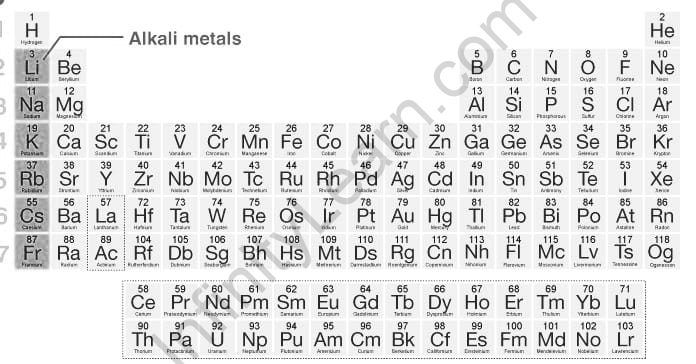
Antacid Metals are extremely receptive and are available as mixtures as they were. They are electropositive metals with unit valence.
Electronic Configuration of Alkali Metals
- Antacid metals have one electron in their valence shell.
- The electronic setup is given by ns1. For instance, the electronic setup of lithium is given by 1ns1 2ns1.
- They will quite often lose the external shell electron to shape cations with charge +1 (monovalent particles).
- This makes them the most electropositive components and because of similar explanations, they are not found in the unadulterated state.
Patterns in Physical Properties of Alkali Metals
Down the segment, the atomic charge increments and a new orbital gets added to every antacid iota. Here, we have examined a few significant patterns in the actual properties of salt metals as we go down the segment.
Expanding request of Atomic and Ionic Radius: Li ˂ Na ˂ K ˂ Rb ˂ Cs and Li+ ˂ Na+ ˂ K+ ˂ Rb+ ˂ Cs+
The thickness of Alkali Metals
Having the biggest range and volume, antacid components have the most reduced thickness. So they are extremely delicate and can be cut with a blade. Lithium, sodium and potassium are lighter than water. Potassium has the least thickness among antacid metals.
FAQs
Q: What are the properties of salt metals?
Ans: The salt metals are a gathering of substance components in the intermittent table with the accompanying physical and compound properties:
- gleaming.
- delicate.
- brilliant.
- profoundly receptive at standard temperature and strain.
- promptly lose their furthest electron to frame cations with a charge of +1.
What are the normal purposes of salt metals?
Modern applications incorporate hotness safe glass and ceramics, lithium oil oils, transition added substances for iron, steel and aluminum creation. Cell phones and electric vehicles rely upon lithium-particle batteries.
Where are antacid metals found?
There are salt metals surrounding you at the present time. Sodium is seen as in table salt, lithium in your telephone battery and potassium in your bananas. Salt metals make up six unique components found in the primary segment of the intermittent table





