Table of Contents
CBSE Previous Year Question Papers Class 12 Chemistry 2014 Outside Delhi
Time allowed: 3 hours
Maximum Marks: 70
CBSE Previous Year Question Papers Class 12 Chemistry 2014 Outside Delhi Set I
Question 1.
What is the effect of temperature on chemisorption? [1]
Answer:
Chemisorption initially increases then decreases with rising in temperature. The initial increase is due to the high energy of activation and the decrease afterwards is due to the exothermic nature of adsorption equilibrium.
Question 2.
What is the role of zinc metal in the extraction of silver? [1]
Answer:
Zinc is used as a reducing agent to recover silver from its cyanide complex. It reduces Ag+ to Ag and itself get oxidised to Zn2+.
Question 3.
What is the basicity of H3PO3? [1]
Question 4.
Identify the chiral molecule in the following pair: [1]
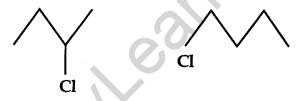
Answer:
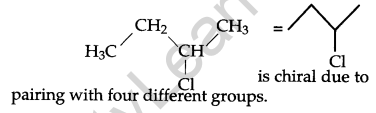
Question 5.
Which of the following is a natural polymer? [1]
Buna-S, Proteins, PVC Economics type
Answer:
Protein is a natural polymer having amino acid as a monomer.
Question 6.
The conversion of primary aromatic amines into diazonium salts is known as ____ [1]
Answer:
Diazotisation reaction.
Question 7.
What are the products of hydrolysis of sucrose? [1]
Answer:
The products of Hydrolysis of sucrose are: Glucose and Fructose
Question 8.
Write the structure of p-methylbenzaldehyde.
Answer:
![]()
Question 9.
An element with density 2.8 g cm-3 forms an f.c.c. unit cell with edge length 4 × 10-8 cm. Calculate the molar mass of the element. (Given NA = 6.022 × 1023 mol-1) [2]
Question 10.
- What type of non-stoichiometric point defect is responsible for the pink colour of LiCl?
- What type of stoichiometric defect is shown by NaCl? [2]
OR
How will you distinguish between the following pairs of terms?
- Tetrahedral and octahedral voids
- Crystal lattice and unit cell
Question 11.
State the Kohlrausch law of independent migration of ions. Why does the conductivity of a solution decrease with dilution? [2]
Answer:
It states that the limiting molar / Conductivity of an electrolyte can be expressed as a sum of the individual contribution of anion and cation of the electrolyte.
With dilution, the number of ions per unit volume of electrolytes decreases and hence, conductivity decreases.
Question 12.
For a chemical reaction R → P, the variation in the concentration (R) Vs. time (t) plot is given as [2] (i)
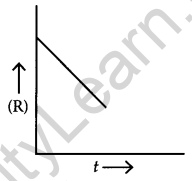
- Predict the order of the reaction.
- What is the slope of the curve?
Answer:
- Zero-order reaction
- The slope of the curve is (-K). i.e., a negative slope.
Question 13.
Explain the principle of the method of electrolytic refining of metals. Give one example. [2]
Answer:
In the electrolytic method, the impure metal is made anode. A strip of the same metal in pure form is used as the cathode. They are put in a suitable electrolytic bath containing soluble salt of the same metal. The more basic metal remains in the solution and the less basic ones go to the anode mud.
For e.g.: Copper is refined using an electrolytic method, the net result of electrolysis is the transfer of copper in pure form from the anode to the cathode.
Anode: Cu → Cu2+ + 2e–
Cathode: Cu2+ + 2e– → Cu
Question 14.
Complete the following equations: [2]
- P4 + H2O →
- XeF4 + O2F2 →
Answer:
- P4 + H2O → H3PO4 + H2
- XeF4 + O2F2 → XeF6 + O2
Question 15.
Draw the structures of the following:
(i) XeF2
(ii) BrF3 [2]
Answer:
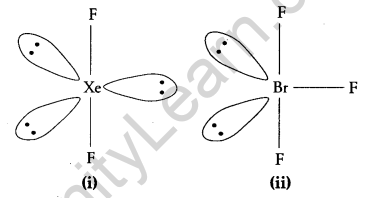
Question 16.
Write the equations involved in the following reactions: [2]
(i) Reimer-Tiemann reaction
(ii) Williamson synthesis
Answer:
(i) Reimer Tiemann reaction:
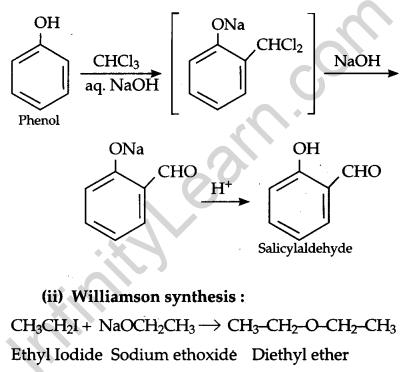
Question 17.
Write the mechanism of the following reaction:
![]()
Answer:
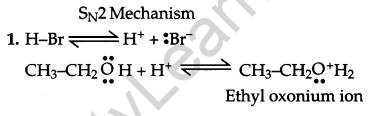
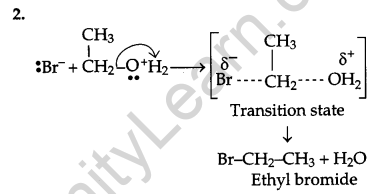
Question 18.
Write the name of monomers used for getting the following polymers: [2]
- Bakelite
- Neoprene
Answer:
- Phenol and formaldehyde are the monomers used for the formation of Bakelite.
- Chloroprene is the monomer used to prepare Neoprene.
Question 19.
(a) Calculate ΔrG0 for the reaction
Mg(s) + Cu2+(aq) → Mg2+(aq) + Cu(s)
Given: E0cell = 2.71 V, 1F = 96500 C mol-1
(b) Name the type of cell that was used in Apollo space program for providing electrical power. [3]
Answer:
(a) Mg(s) + Cu2+(aq) → Mg2+(aq) + Cu(s)
ΔrG° = – nFE°cell
= -2 × 96500 × 2.71
= -523.03 kj mol-1
(b) Hydrogen-oxygen fuel cells and solar cells.
Question 20.
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume:
SO2Cl2 (g) → SO2(g) + Cl2 (g)

Calculate the rate constant. (Given: log 4 = 0.6021, log 2 = 0.3010) [3]
Answer:
SO2Cl2 (g) → SO2(g) + Cl2 (g)
dt1 t = 0, P0 ….. 0
At t = t, P0 – P …….. P
The total pressure of the thermal decomposition of SO2Cl2 time t.
Pt = (P0 – P) + P + P
Pt = P0 + P
Hence, P = Pt – P0
P0 – P = P0 – (Pt – Po) = 2P0 – Pt

Question 21.
What are emulsions? What are their different types? Give one example of each type. [3]
Answer:
Emulsions are colloids in which both the dispersed phase and dispersing medium are liquid. It is a mixture of two or more liquids that are normally immiscible. They are of two types:
(i) Oil in water type emulsions (O/W): In such emulsions, oil is the dispersed phase and water is the dispersed medium.
e.g., Milk, Vanishing cream.
(ii) Water in oil type emulsions (W/O): In such emulsions, water is the dispersed phase and oil is the dispersed medium.
e.g., Butter, Cod liver oil.
Question 22.
Given the reasons for the following:
(i) (CH3)3 P = O exists but (CH3)3N = O does not.
(ii) Oxygen has less electron gain enthalpy with a negative sign than sulphur.
(iii) H3PO2 is a stronger reducing agent than H3PO3. [3]
Answer:
(ii) Due to a small size and high electronegativity of oxygen compared to sulphur, oxygen has less electron gain enthalpy.
Question 23.
(i) Write the IUPAC name of the complex [Cr(NH3)4Cl2]Cl.
(ii) What type of isomerism is exhibited by the complex [Co(en)3]3+? (en = ethane-1,2-diamine)
(iii) Why is [NiCl4]2- paramagnetic but [Ni(CO)4] is diamagnetic [3]
(At. nos.: Cr = 24, Co = 27, Ni = 28)
Answer:
(i) Tetraammine dichloridochromium(III) chloride
(ii) Optical isomerism is shown by the complex [CO(en)3]3+
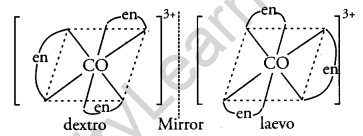
(iii) [Ni(Cl)4]2-, Ni is in +2 oxidation state with the electronic configuration 3d84s0. As Cl– is a weak ligand it cannot pair up the electrons in 3d orbitals therefore, [NiCl4]2- is paramagnetic. In [Ni(CO)4], Ni is in 0 oxidation state with the electronic configuration 3d84s2. Co is a strong ligand it causes 4s electrons to shift to 3d and pair up 3d electrons. Therefore, Ni(CO)4 is diamagnetic.
Question 24.
(a) Draw the structure of major moon halo products in each of the following reactions:
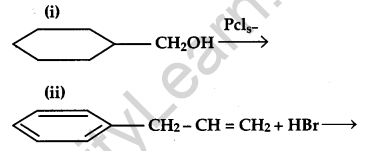
(b) which halogen compound in each of the following pairs will react faster in SN2 reaction:
(i) CH3Br or CH3I
(ii) (CH3)3C-Cl or CH3-Cl
Answer:
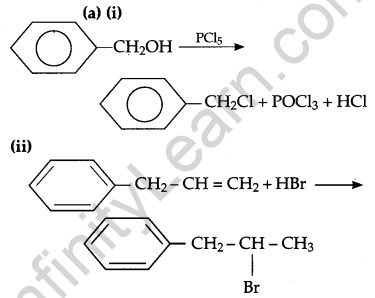
(b) (i) CH3-I will react faster because Iodine (I) is a better leaving group.
(ii) CH3-CI will react faster because it is a primary halide and it undergoes SN2 reaction faster.
Question 25.
Account for the following:
(i) Primary amines (R-NH2) have a higher boiling point than tertiary amines (R3N).
(ii) Aniline does not undergo Friedel-Crafts reaction.
(iii) (CH3)2NH is more basic than (CH3)3N in an aqueous solution. [3]
OR
Give the structures of A, B and C in the following reactions:
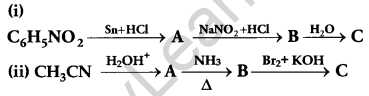
Answer:
(i) Due to intermolecular hydrogen bonding in primary amines (presence of more number of H-atoms). They have a high boiling point in comparison to tertiary amines.
(ii) Aniline does not undergo Friedel-Crafts reaction because Aniline is basic in nature and reacts with AlCl3 to form a salt. The positive charge on the N-atom, electrophilic substitution in the benzene ring is deactivated.
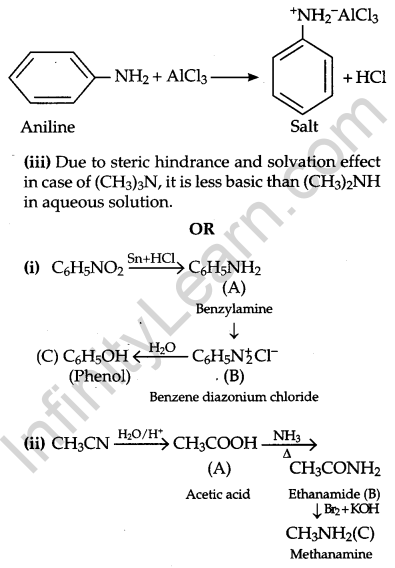
Question 26.
Define the following terms related to proteins: [3]
- Peptide linkage
- Primary structure
- Denaturation
Answer:
- Peptide linkage is the amide linkage formed by -COOH group of one a-amino acid and -NH2 group of other α-amino acids by loss of a water molecule.
- The sequence in which various amino acids are arranged in a linear structure with no intermediate bonding is called the primary structure of a protein.
- When a protein in its native form is subjected to a change, such as a temperature, pressure etc. Due to this protein loses its biological activity and this is called denaturation of a protein e.g., curdling of milk.
Question 27.
On the occasion of World Health Day.
Dr Satpal organized a ‘health camp’ for the poor fanners living in a nearby village. After check¬up, he was shocked to see that most of the farmers suffered from cancer due to regular exposure to pesticides and many were diabetic. They distributed free medicines to them. Dr Satpal immediately reported the matter to the National Human Rights Commission (NHRC). On the suggestions of NHRC, the government decided to provide medical care, financial assistance, setting up of super-speciality hospitals for treatment and prevention of the deadly disease in the affected villages all over India.
(i) Write the values shown by (a) Dr Satpal (b) NHRC?
(ii) What type of analgesics are chiefly used for the relief of pains of terminal cancer?
(iii) Give an example of artificial sweetener that could have been recommended to diabetic patients. [3]
Answer:
(ii) Narcotic analgesics like morphine and heroin.
(iii) Aspartame.
Question 28.
(a) Define the following terms:
(i) Molarity
(ii) Molal elevation constant (kb)
(b) A solution containing 15 g urea (molar mass = 60 g mol-1) per litre of a solution in water has the same osmotic pressure (isotonic) as a solution of glucose (molar mass = 190 g mol-1) in water. Calculate the mass of glucose present in one litre of its solution. [2, 3]
OR
(a) What type of deviation is shown by a mixture of ethanol and acetone? Give reason.
(b) A solution of glucose (molar mass = 180 g mol-1) in water is labelled as 10% (by mass). What would be the molality and molarity of the solution? (Density of solution = 1.2 g mL-1)
Answer:
(a) (i) The number of moles of a solute present in one litre of solution is known as its molarity.
(ii) The elevation in the boiling point of a solution when one mole of non-volatile solute is dissolved in one kilogram of a volatile solvent is known as molal elevation constant (Kb).
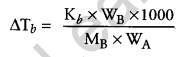
WB = Mass of solute
MB = Molar mass of solute
WA = Mass of solvent.
(b) For isotonic solution,
π1 = π2
C1 = C2 (at same temp.)
or n1 = n2 (at same Vol.)
\(\frac { 15 }{ 60 }\) = \(\frac { x }{ 180 }\)
x = 45 g, mass of glucose per litre of solution.
OR
(a) The mixture of ethanol and acetone shows positive deviation from Raoult’s law. In pure ethanol, hydrogen bond exists between the molecules. On adding acetone to ethanol, acetone molecules get in between the molecules of ethanol thus breaking some of the hydrogen bonds and weakening the molecular interactions this leads to an increase in vapour pressure resulting in a positive deviation from Raoult’s law.
(b) Mass of glucose = 10 g
Mass of solution = 100 g
Mass of water = 100 – 10 = 90 g

Question 29.
(a) Complete the following equations:
(i) Cr2O72- + 2OH– →
(ii) MnO4– + 4H+ + 3e– →
(b) Account for the following:
(i) Zn is not considered as a transition element.
(ii) Transition metals form a larger number of complexes.
(iii) The E value for the Mn3+/Mn2+ couple is much more positive than that for C3+/Cr2+ couple. [2, 3]
OR
(i) With reference to structural variability and chemical reactivity, write the difference between lanthanoids and actinoids.
(ii) Name a member of the lanthanoid series which is well known to exhibit +4 oxidation state.
(iii) Complete the following equation :
MnO4– + 8H+ + 5e– →
(iv) Out of Mn3+ and Cr3+, which is more paramagnetic and why? (atomic nos : Mn = 25, Cr = 24) [5]
Answer:
(a) (i) Cr2O72- + 2OH– → 2CrO42- + H2O
(ii) MnO4– + 4H+ + 3e– → MnO2 + 2H2O
(b) (i) Zinc has 3d104s2 configuration with no unpaired d-orbital electron and hence it is not considered as a transition element.
(ii) Transition metals have vacant orbitals to accommodate lone pairs of electrons for bond formation and have high charge density, therefore, they form complexes.
(iii) Due to the high stability of Mn2+ (due to its half-filled 3d subshell) than Mn3+ while Cr3+ is more stable than Cr2+.
OR
(i)
| S.No. | Actinoids | Lanthanoids |
| 1. | Actinoids have a stronger tendency to form complexes. | Lathanoids formless complexes. |
| 2. | They show more number of oxidation states. | They show less number of oxidation states. |
| 3. | They are radioactive. | Lanthanoids except promethium is not radioactive. |
(ii) Cerium (Ce) is a lanthanoid element, which is well known to exhibit a +4 oxidation state.
(iii) MnO–4 + 8H+ + 5e– → Mn2+ + 4H2O
(iv) Mn3+ is more paramagnetic as it has four impaired electrons while Cr3+ has only three.
Mn2+ = [Ar] 4s03d4
Cr3+ = [Ar] 4s03d3
Question 30.
(a) Write the products formed when CH3CHO reacts with the following reagents:
(i) HCN
(ii) H2N-OH
(iii) CH3CHO in the presence of dilute NaOH
(b) Give simple chemical tests to distinguish between the following pairs of compounds.
(i) Benzoic acid and Phenol
(ii) Propanal and Propanone. [3, 2]
OR
(a) Account for the following: (2, 2, 1)
(i) Cl–CH2COOH is a stronger acid than CH3COOH.
(ii) Carboxylic acids do not give reactions of the carbonyl group.
(b) Write the chemical equations to illustrate the following name reactions:
(i) Rosenmund reduction
(ii) Cannizzaro’s reaction
(c) Out of CH3CH2-CO-CH3 and CH3CH2-CH2-CO-CH3, which gives iodoform test?
Answer:
(a) (i) On the reaction of acetaldehyde with hydrogen cyanide, it gives 2-Hydroxypropanenitrile as the product.
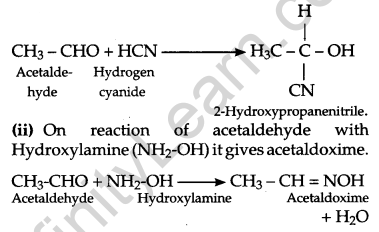
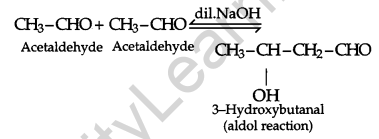
(iii) When 2 molecules of acetaldehyde react with each other in presence of dil. NaOH, 3-Hydroxybutanal is obtained. This is an aldol reaction and further proceeds the reaction when heated.
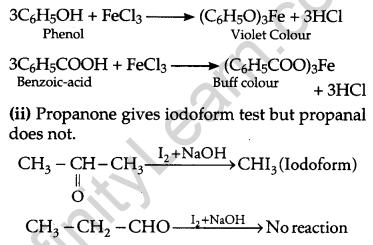
(b) (i) Phenol reacts with FeCl3 to give violet coloured precipitate while benzoic acid gives a buff coloured precipitate.
OR
(a) (i) Because of -I effect of Cl atom in ClCH2COOH and +I effect of CH3 group in CH3COOH the electron density in the O-H bond in ClCH2COOH is much lower than CH3COOH. Hence ClCH2COOH acid is a stronger acid than CH3COOH.
(ii) In carboxylic acids, the carboxyl group is not free as it is involved in resonance
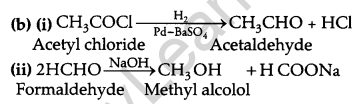
(c) CH3CH2CH2COCH3, being a methyl ketone gives iodoform test.
CBSE Previous Year Question Papers Class 12 Chemistry 2014 Outside Delhi Set II
Note: Except for the following questions, all the remaining questions have been asked in the previous set.
Question 1.
Why is adsorption always exothermic? [1]
Answer:
Adsorption being a surface phenomenon leads to a decrease in surface energy and hence is exothermic in nature.
Question 2.
Name the method used for refining of Nickel. [1]
Answer:
Mond’s process is used for refining of Nickel.
Question 3.
Why does NO2 dimerise? [1]
Question 4.
Based on molecular forces, what type of polymer is neoprene? [1]
Answer:
Elastomer.
Question 5.
What are the products of hydrolysis of maltose? [1]
Answer:
Two molecules of α-D-glucose
Question 6.
Write the structure of 4-chloropentan-2 one. [1]
Answer:
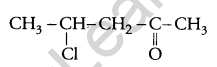
Question 9.
Write the name of monomers used for getting the following polymers: [2]
- Terelyne
- Nylon-6,6
Answer:
- Ethylene glycol and Terephthalic acid.
- Adipic acid and Hexamethylene diamine.
Question 10.
Describe the role of the following: [2]
- SiO2 in the extraction of copper from copper matte.
- NaCN in froth floatation process.
Answer:
- SiO2 (Silica) acts as a flux in the extraction of copper from copper matte to remove ferrous oxide as ferrous silicate slag.
FeO + SiO2 → FeSiO3 (Slag) - NaCN is used as a depressant as it forms zinc complex, Na2[Zn(CN)4] on the surface of ZnS thereby preventing it from forming froth.
Question 11.
Complete the following equations: [2]
(i) Ag + PCl5
(ii) CaF2 + H2SO4
Answer:
(ii) CaF2 + H2SO4 → CaSO4 + 2HF
Question 12.
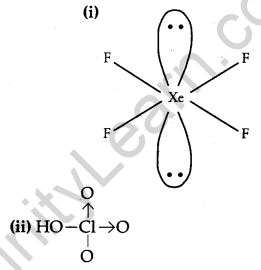
Draw the structures of the following: [2]
(i) XeF4
(ii) HClO4
Answer:
Question 13.
(i) Write the type of magnetism observed when the magnetic moment is oppositely aligned and cancel out each other.
(ii) Which stoichiometric defect does not change the density of the crystal? [2]
Question 14.
Define the following terms: [2]
- Fuel cell
- Limiting molar conductivity (∧m°)
Answer:
- A fuel cell is a device that converts chemical energy from the combustion of a fuel into electric energy through a chemical reaction.
- Molar conductivity of electrolyte at infinite dilution or when concentration approaches zero is called limiting molar conductivity. It is expressed as ∧m°
Question 19.
Define the following terms: [3]
(i) Glycosidic linkage
(ii) Invert sugar
(iii) Oligo saccharides
Answer:
(i) The linkage between two monosaccharide units through oxygen atom is known as glycosidic linkage.
(ii) Sucrose is called invert sugar as it produces equimolar quantities of glucose and fructose on hydrolysis it gives an equimolar mixture of D – (+)-glucose, and D – (-)-fructose, which is laevorotatory. This change of specific rotation from dextrorotation to laevorotation is called inversion of sugar and the mixture so obtained is called invert sugar.
(iii) Carbohydrate which on hydrolysis give two to ten molecules of monosaccharides is called oligosaccharide e.g. sucrose.
CBSE Previous Year Question Papers Class 12 Chemistry 2014 Outside Delhi Set III
Note: Except for the following questions, all the remaining questions have been asked in previous sets.
Question 1.
What are the dispersed phase and dispersion medium in milk? [1]
Answer:
The dispersed phase is oil or fat and the dispersion medium is water.
Question 2.
Name the method used for refining of copper metal. [1]
Answer:
Electrolytic refining.
Question 3.
Why does NH3 act as a Lewis base? [1]
Answer:
Due to the presence of lone pair on nitrogen in NH3, It can donate its lone pair of electrons and it forms coordinate bonds with Lewis acids and acts as Lewis base.
Question 5.
Which of the following is a fibre? [1]
Nylon, Neoprene, PVC
Answer:
Nylon
Question 6.
Write the products of hydrolysis of lactose. [1]
Answer:
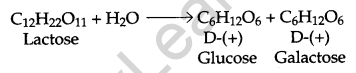
Question 8.
Write the structure of 2-hydroxybenzoic acid. [1]
Answer:
Question 9.
Complete the following equations: [2]
- Cu + 2H2SO4 (Cone.) →
- XeF2 + H2O →
Answer:
- Cu + conc. 2H2SO4 → CuSO4 + SO2 + 2H2O
- 2XeF2 + 2H2O → 2Xe + 4HF + O2
Question 10.
Draw the structure of the following: [2]
(i) XeO3
(ii) H2SO4
Answer:
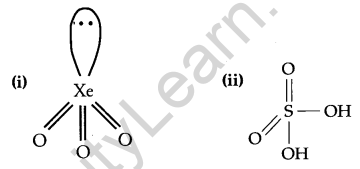
Question 11.
Write the name of monomers used for getting the following polymers: [2]
- Teflon
- Buna-N
Answer:
- Tetrafluoroethylene
- 1, 3-Butadiene and Acrylonitrile
Question 13.
(i) Write the type of magnetism observed when the magnetic moment is aligned in parallel and anti-parallel directions in unequal numbers. [2]
(ii) Which stoichiometric defect decreases the density of the crystal?
Question 14.
Define the following terms:
- Molar conductivity (∧m)
- Secondary batteries [2]
Answer:
- Molar conductivity is defined as the conducting power of all the ions produced by dissolving one mole of an electrolyte in solution.
∧m = \(\frac { K }{ C }\) - Secondary batteries are those batteries which can be recharged by passing an electric current through them and hence can be used over again e.g. Lead storage battery.
Question 17.
Write the principle behind the froth floatation process. What is the role of collectors in this process? [2]
Answer:
Froth floatation method has been in use for removing gangue from sulphide ores. In this process, a suspension of the powdered ore is made with water.
Collectors (e.g. pine oils, fatty acids, etc.) enhance non-wettability of the mineral particles and used to skim the froth off the surface.
Question 23.
Define the following terms : [3]
(i) Nucleotide
(ii) Anomers
(iii) Essential amino acids
Answer:
(i) A Nucleotide contains all the three basic components of nucleic acid i.e, a pentose sugar, a nitrogenous base and a phosphoric add. When nucleoside is linked to phosphoric add at 5′ position of the sugar moiety, we get a nucleotide.
(ii) The carbohydrate which differs in configuration at the glycosidic carbon (i.e., C1 in aldoses and C2 in ketoses) are called anomers. e.g. α-D-(+) glucose and β-D-(+) glucose.
(iii) Essential amino acids are those amino acids which cannot be synthesised by the body and need to be consumed through diet. eg. Valine.



