
Courses

By Shailendra Singh
|
Updated on 10 Jan 2025, 11:56 IST
The sector of interstitial compounds is mature, with a large body of literature. Nonetheless, a fundamental question such as the nature of their electronic structure is still the subject of lively (and sometimes heated) debate today. Furthermore, the industry has only scratched the surface of these materials’ myriad technical possibilities. Each year, people produce tones of the interstitial compound tungsten carbide for use in grinding and cutting tools, primarily due to its high hardness. Other properties, on the other hand, have seen little commercial application as of yet. To name a few, they are refractory while remaining metallic, and they frequently have abnormally low work functions, making them excellent electron emitters.
This combination suggests applications such as magnetohydrodynamic channel electrodes. Some interstitial compounds, like NBN, are superconducting, with transition temperatures second only to Nb3Sn. Tungsten carbide and related chemicals are moderately good catalysts that can operate in chemically hostile environments, such as those found in fuel cells. Many of them are good ferro and antiferromagnets. As we will try to demonstrate, this class of compounds offers both fascinating possibilities for technologists and real challenges for researchers.
We realise that transition metals are the elements found in the periodic table’s d-block. Cobalt, nickel, iron, and other transition metals are examples. Transition metals can form complex non-stoichiometric compounds. These are compounds with arbitrary structure and proportions. Fe0.94O, for example, has a structure that is primarily due to the variable valency of transition elements.
Non-stoichiometry is caused by defects in solid structures as well as the variable oxidation state of transition metals. These compounds do have a variable composition and are formed due to the variability of oxidation states and solid defects. Interstitial and nonstoichiometric compounds are sometimes the same.
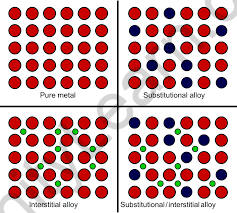
These are transition metal compounds formed when small atoms such as H, C, or N are trapped inside the interstitial vacant spaces in the metal’s crystal lattices. A few examples of interstitial compounds are TiC, TiH1.73, Mn4N, Fe3H etc.
Interstitial compounds were first proposed in the late 1930s and are commonly referred to as Hagg phases after Hägg. Transition metals typically crystalise in either hexagonal close-packed or face-centered cubic structures, both of which are composed of layers of hexagonally close-packed atoms. There are two types of interstices, or holes, in both of these very similar lattices:

An analysis of transition temperature measurements of pseudo-binary and ternary interstitial alloys reveals that there is no simple direct relationship between transition temperature and quantities such as valence electron-atom ratio, mass, and lattice volume. We have successfully systematised the experimental data into a general empirical model that can predict the most promising materials for achieving high transition temperatures.
The transition elements combine to form a wide range of interstitial compounds. Small atoms such as hydrogen, carbon, boron, and nitrogen fill the empty spaces in these compounds’ lattices. The small atoms enter the voids or interstitial sites between the crystalline metal’s packed atoms. We can say that, they are typically non-stoichiometric and neither ionic nor covalent. The expressions of non-stoichiometric compounds do not correspond to any of the metal’s normal oxidation states. These compounds are known as interstitial compounds because of the unique properties of their composition. These compounds are harder than pure metals and have higher melting points. They are chemically inert and retain metallic conductivity. The presence of small atoms reduces the malleability and ductility of metals while increasing their tensile strength.
Interstitial compounds also can have semi conductivity, fluorescence, and act as heterogeneous catalysts. Their catalytic activity is associated with the variable oxidation states of d-block elements and their compounds, as well as their ability to form interstitial compounds that can absorb and activate the reacting species.
Interstitial compounds can be formed by elements from the 3d-transition series, such as Ti2C, V2C, ScN, TiN, Fe4N and so on. These compounds have alloy properties such as hardness and conductivity. These elements can also combine to form non-stoichiometric compounds.

Generally, an interstitial compound also known as an interstitial alloy, is a compound formed when an atom with a small enough radius sits in a metal lattice’s interstitial “hole.” Small atoms include hydrogen, boron, carbon, and nitrogen.
They are tough and have excellent heat and electricity conductors. They have the same chemical properties as the parent metal. They have higher melting points than pure metals.
Non – stoichiometric compounds are said to be chemical compounds that deviate from stoichiometry in the sense that their elemental composition cannot be represented by a ratio of well-defined natural numbers and thus violate the law of definite proportions