To Set up Simple Daniell Cell and Determine its emf
Theory
When a copper electrode dipped in copper sulphate solution is connected to a zinc electrode dipped in the zinc sulphate solution, then electrons flow from zinc electrode to copper electrode and the chemical reactions take place as :
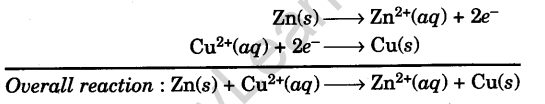
Apparatus and Chemicals
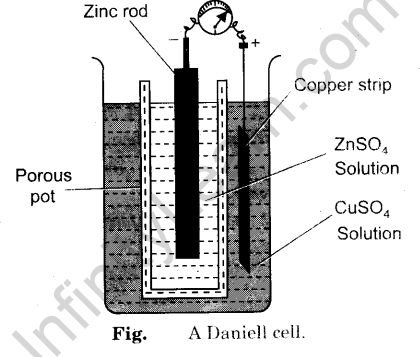
One beaker, a porous pot, connecting wires, milli voltmeter, sand paper, zinc strip, copper strip, 1 M ZnS04 solution and 1 M CuS04 solution.
Procedure
- Take copper sulphate solution in a clean beaker.
- Clean the copper strip with the help of sand paper and dip it into copper sulphate solution.
- Take zinc sulphate solution in a porous pot.
- Clean the zinc strip with the help of sand paper and dip it into zinc sulphate solution.
- Keep the porous pot in the beaker.
- Connect the copper strip with the positive terminal and zinc strip with the negative terminal of a voltmeter as shown in Fig.
- Note the position of the pointer in the voltmeter and record the reading in your notebook.
Observation
The emf of the Daniell cell is volts.
Precautions
- The concentration of copper sulphate and zinc sulphate should neither be too low nor too high.
- The porous pot should not be completely dipped into the copper sulphate solution, i.e., the copper sulphate solution should not be allowed to enter into the porous pot.
- Clean zinc and copper strips with sand paper before use.
- Carry out dilution of the solution carefully.
- Note the reading only when the pointer becomes stable.
- Connect copper strip with the positive terminal of voltmeter and zinc strip with negative terminal.


