Table of Contents
Nature of Carbonyl Group: A carbonyl group is a chemically organic functional group composed of a carbon atom double-bonded to an oxygen atom –> [C=O] The most superficial carbonyl groups are aldehydes and ketones usually attached to another carbon compound. These structures can be planted in multiplex sweet mixtures contributing to smell and taste. There are two main types of carbonyl compounds: organic and inorganic. Here, we will discuss organic carbonyl compounds.
Nature Of Carbonyl Group
The carbonyl group is the organic functional group composed of the carbon atom that has a double bond with the oxygen atom. Ketones and aldehydes are the most superficial carbonyl groups, and usually, they are attached to another carbon compound. In any perfumed mixture, their structure can be planted, and they have a significant donation to the taste and the smell of the scented composites. The situation where carbon makes the double bond with the oxygen is considered the carbonyl group entity, and the group members are known as the carbonyl compounds. Generally, the carbon and oxygen of the carbonyl group are planar and sp2 hybridised.
The carbon-oxygen bond is polarised due to the high electronegativity difference between carbon and oxygen tittles of the> C =O bond. Oxygen has an advanced electronegativity compared to that of the carbon particle. Wherefore, the carbonyl carbon demonstrates the property of a Lewis acid, whereas the carbonyl oxygen demonstrates the property of a Lewis base.
Carbonyl compounds contain significant dipole moments. Therefore, it demonstrates more polarity than ethers. The high opposition of the carbonyl group is substantially due to the resonance concerning neutral and dipolar structures.
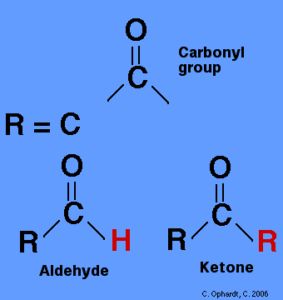
Introduction of Carbonyl Compounds
Carbonyl Compounds definition can be given as the compounds that hold a carbonyl group (the C=O group) are known as carbonyl compounds.
The carbonyl grouping is presumably the essential functional group of organic chemistry. These are an integral part of organic chemistry, and their primary members are called ketones, carboxylic acids, and aldehydes.
Carbonyl mixtures are further divided into organic and inorganic carbonyl combinations. This complete article details the organic carbonyl compounds. Carbonyl groups are the functional groups of aldehydes and ketones. Carbonyl groups are located at the end of aldehydes, while carbonyl groups are located in the middle of ketones. Molecular rotation is possible because of the double bond of carbonyl groups. Nature contains various types of carbonyl compounds. The acyl groups (C-R = O) all have a substituent attached. R consists of an alkyl, an alkenyl, an alkynyl, or a combination of these functional groups.
Also Read: Aldehydes, Ketones and Carboxylic Acids Class 12 Notes Chemistry Chapter 12
Examples of Carbonyl Compounds
Carbamates, urea, acyl chlorides, chloroformates, carbonate esters, lactones, thioesters, lactams, isocyanates, and hydroxamates.
Types of metal carbonyls
Metal carbonyls are cooperation complexes of transition matter with carbon monoxide ligands. The general formula of material carbonyls is Mx (CO) y. A lone couple of electrons are available on both carbon and oxygen tittles of a carbon monoxide ligand. As the carbon titles present electrons to the essence, these complexes are carbonyls.
I. Directly Using the ‘CO ‘.
Mononuclear essence carbonyls similar as Fe (CO) 2, Ni (CO)4, and binuclear essence carbonyls similar to Co2 (CO) 8 may be prepared by the direct response of CO with finely divided essence at suitable temperature and pressure.
The main requirement of this method is that the metal centre must be in a reduced low oxidation state to facilitate CO binding to the metal centre through metal to ligand π–back donation.
II. Reductive Carbonylation
Treating salts like Ru(acac)3, CrCl3, VCl3, CoS, Col2 with carbon monoxide in the presence of suitable reducing agents like Mg, Ag, Cu, Na, H2, LiAlH4, etc. gives metal carbonyls.
III. From Mononuclear Carbonyls
When a cold answer of Fe (CO) 5/ naughts (CO) 5 in numbing acetic acid CH3COOH is irradiated with ultraviolet light, Fe2 (CO) 9/ Os2 (CO) 9 are carried.
FAQs on Nature of Carbonyl Group
Many carbonyl compounds are aromatic. Substances that include carbonyls include the chemical responsible for the smell of vanilla and cinnamon. Formaldehyde, another carbonyl structure, is generally used for embalming in interring homes.
Carbonyl groups are reduced by reacting with hydride reagents like LiAlH4 and NaBH4, catalytic hydrogenation, or the baker's yeast. Ketones form secondary alcohols, while esters, carboxylic acids, and aldehydes include primary drinks. what is used for carbonyl?
What is carbonyl reduction?



