Table of Contents
Chemistry – Chapter 14
Get CBSE Notes for Class 11 Chemistry on Infinity Learn for free.
Environmental Chemistry
• Environmental Chemistry
It is the branch of science that deals with the chemical changes in the environment. It includes our surroundings as air, water, soil, forest etc.
• Environmental Pollution
It is the effect of undesirable changes in our surroundings that have harmful effects on plants, animals and human beings.
• Pollutants
A substance, which causes pollution, is known as a pollutant. Pollutants can be solid, liquid or gaseous substances. Present in higher concentration, it can be produced due to human activities or natural happenings.
• Troposphere
The lowest region of the atmosphere, in which human beings along with other organisms live, is called the troposphere.
It extends to the height of about 10 km from sea level. It contains air, water vapours, clouds etc. The pollution in this region is caused by some poisonous gases, smoke fumes, smog etc.
• Stratosphere
It extends from a height of 10 to 50 km above sea level. Ozone and some other gaseous substances present in this region are responsible for the pollution.
• Tropospheric Pollution
Pollution in this region is caused by the presence of undesirable gaseous particles like oxides of sulphur, nitrogen and carbon, hydrocarbons along with solid particles like dust, mist, fumes, smoke etc.
• Oxides of Sulphur
These are produced when coal containing sulphur is burnt.
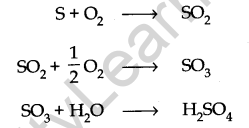
It is also produced during volcanic eruptions.
Harmful effects:
(i) It is poisonous to both animals and plants.
(ii) A very high concentration of S02 may cause respiratory diseases e.g., asthma,bronchitis, emphysema in human beings.
(iii) It causes irritation to the eyes, resulting in tears and redness.
(iv) Its high concentration leads to the stiffness of flower buds.
(v) Particulate matter present in the air can catalyse the formation of sulphur trioxide from sulphur dioxide.
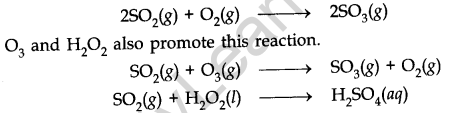
• Oxides of Nitrogen
The main oxides of nitrogen are nitric oxide (NO) and nitrogen dioxide (NO2).
Major Sources:
(i) Lightning discharge results in the combination of N2 and 02 to form NO.
(ii) Combustion of gasoline in automobiles, burning of hydrocarbons and coal etc.
Harmful effects:
Nitric oxide itself is not harmful to human beings, but it is very unstable and changes to nitrogen dioxide which is toxic in nature. These effects are as follows:
(i) It reacts with Ozone (03) present in the atmosphere and thus decreases the density of Ozone.
(ii) It affects the respiratory system and damages the lungs.
(iii) Higher concentrations of N02 damage the leaves of plants and retard the rate of photosynthesis.
(iv) It causes cracks in the rubber.
(v) Nitrogen dioxide is also harmful to various textile fibres and metals.
• Hydrocarbons
Incomplete combustion of fossil fuel in industry and thermal power plants and the exhaust of automobiles release hydrocarbons into the atmosphere constantly causing pollution. Harmful Effects:
(i) They cause cancer.
(ii) Methane is one of the greenhouse gases.
(iii) They harm plants in various ways like breakdown of tissues, shedding of leaves etc.
• Oxides of Carbon
Carbon dioxide:
0.03% C02 is present in air by Volume.
Major Sources:
(i) By the burning of fossil fuels.
(ii) By the decomposition of limestone during the manufacture of cement.
(iii) Emitted during volcanic eruptions.
(iv) C02 is released into the atmosphere by respiration.
Harmful effects:
Deforestation and the burning of fossil fuels increase the C02 level which is mainly responsible for global warming.
Carbon Monoxide: Carbon Monoxide is a colourless and odourless gas.
Major Sources:
(i) Released by the automobile exhaust.
(ii) Incomplete combustion of coal, fire wood, petrol etc.
(iii) By the dissociation of C02 at high temperature.
Harmful effects:
(i) It binds to haemoglobin to form carboxyhaemoglobin which is more stable than the oxygen-haemoglobin complex. Its concentration in blood when reaches 3-4%, the oxygen-carrying capacity of blood is greatly reduced.
The oxygen deficiency results in headaches, weak eyesight, nervousness etc.
(ii) It has harmful effects on plants when its concentration is (100 ppm or more).
• Global Wanning and Greenhouse Effect
Greenhouse Effect:
Some gases like carbon dioxide, methane, ozone, water vapours, CFCs have the capacity to trap some of the heat radiations that are released from the earth or from the sun. These gases are known as greenhouse gases and the effect is called the greenhouse effect. This leads to global warming.
Consequences of global warming:
(i) It leads to the melting of polar ice caps and flooding of low lying areas all over the earth.
(ii) Global rise in temperature increases the incidence of infectious diseases like dengue, malaria, yellow fever, sleeping sickness etc.
• Acid Rain
When the pH of the rainwater drops below 5.6, it is known as acid rain.
Normal rain is slightly acidic due to the dissolution of atmospheric carbon dioxide in water.
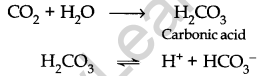
Oxides of nitrogen and sulphur released as a result of the combustion of fossil fuels dissolve in water to form nitric acid and sulphuric acid.
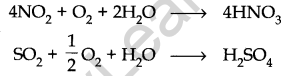
Harmful Effects of Acid Rain:
(i) It has harmful effects on trees and plants as it dissolves and washes away nutrients needed for their growth.
(ii) It has a very bad effect on the aquatic ecosystem.
(iii) Acid rain damages buildings and other structures made of stone or metal. Taj Mahal in India has been affected by acid rain.
![]()
• Particulate Pollutants
Viable Particulates: They are minute living organisms that are dispersed in the atmosphere. e.g., bacteria, fungi) moulds, algae etc.
Non Viable Particulates:
(i) Smoke: It is the mixture of solid and liquid particles formed during the combustion of organic matter,
Example: Cigarette smoke, smoke from the burning of fossil fuel.
(ii) Dust: Composed of fine solid particles (over 2gm in diameter).
It is produced during crushing, grinding and attribution of solid particles.
(iii) Mist: These are produced due to the spray of liquids like herbicides and pesticides over the plants. They travel through the air and form a mist.
(iv) Fumes: They are generally released to the atmosphere by metallurgical operations and also by several chemical reactions.
Harmful Effects of Particulate Pollutants:
(i) Fine particles less than 5 microns penetrate into the lungs. Inhalation of such particles can lead to serious lung diseases including lung cancer.
(ii) Suspended particles of bigger size can hinder the sun rays from reaching the earth surface. This can lower the temperature of the earth and make the weather foggy.
• Smog
This is the common form of air pollution which is a combination of smoke and fog.
Smog exists in two types:
(i) Classical Smog: Occurs in a cool humid climates. It contains smoke, fog and sulphur dioxide. It is also called reducing smog.
(ii) Photochemical Smog: This type of smog results from the action of sunlight on unsaturated hydrocarbons and nitrogen oxides released by vehicles and industries. It has a high concentration of oxidising agents and is, therefore, called oxidising smog.
Formation of Photochemical Smog
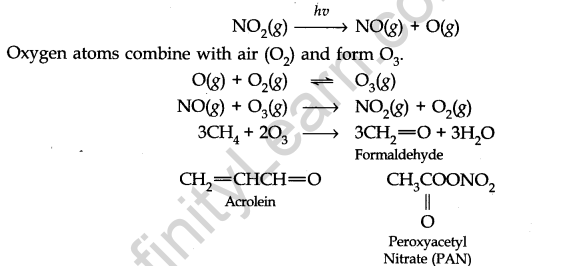
Harmful effects of photochemical smog:
(i) It can cause cough, bronchitis, irritation of the respiratory system etc.
– To control this type of pollution the engines of automobiles are fitted with catalytic converters to check the release of both oxides of nitrogen and hydrocarbons in the atmosphere.
– Some plants like Vitis, Pinus, Juniperus, Quercus, Pyrus can metabolise nitrogen
oxide and therefore, their plantation can be done.
• Stratospheric Pollution
Formation of Ozone: Ozone in the stratosphere is produced by UV radiations. When UV – radiations act on dioxygen (02) molecules, Ozone is produced.
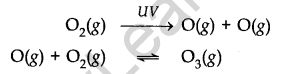
Ozone is thermodynamically unstable and decomposes to molecular oxygen. Thus there exists an equilibrium between the production and decomposition of Ozone molecules.
Depletion of Ozone layer: Ozone blanket in the upper atmosphere prevents the harmful UV radiations from reaching the earth.
But in recent years, there have been reports of depletion of this layer due to the presence of , certain chemicals in the stratosphere. Chlorofluorocarbons (CFCs), nitrogen oxides, chloride, CCl4 etc. are the chemicals responsible for depletion.
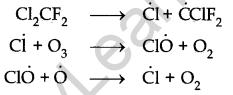
Chlorofluorocarbons dissociate in the presence of light gives chlorine-free radicals which catalyse the conversion of ozone into oxygen.
Effects of the depletion of the Ozone layer:
(i) This leads to many diseases like skin cancer, sunburn, ageing of the skin, cataract etc.
(ii) UV radiations can kill many phytoplanktons, damage fish productivity.
(iii) It can decrease the moisture content of the soil by increasing the evaporation of surface water.
(iv) UV radiations can damage paints and fibres, causing them to fade faster.
• Water Pollution
The presence of undesirable materials in water which is harmful to human beings and plants is known as water pollution. The normal properties of the water can be changed by the presence of these foreign materials.

Causes of Water Pollution:
(i) Pathogens: Pathogens are the bacteria and the other organisms that enter the water from domestic sewage and animal excreta.
Human excreta contain bacteria such as Escherichia coli and Streptococcus faecalis. It causes gastrointestinal diseases.
(ii) Organic Wastes: Organic matter such as leaves, grass, trash etc. can pollute water.
– Excessive growth of phytoplankton within water also pollute water.
– Large numbers of bacteria in water can consume oxygen dissolved in water by decomposing organic matter present in water.
– If the concentration of dissolved oxygen in water is below 6 ppm, the growth of fish gets inhibited.
– If too much organic matter is added to water, all the available oxygen is used up. This can cause the death of aquatic life.
• BOD (Biochemical Oxygen Demand)
It is defined as the amount of oxygen required by bacteria for the breakdown of the organic matter present in a certain volume of a sample of water.
The amount of BOD in water is a measure of the amount of organic material in the water. Clean water has a BOD value of less than 5 ppm.
Highly polluted water could have a BOD value of 17 ppm or more.
• Chemical Pollutants
(i) Industrial Wastes: Chemical reactions carried in the industrial units also pollute water to a great extent. For example, lead, mercury, nickel, cobalt etc. These chemicals give a very bad effect on the groundwater and water bodies that are polluted due to the chemical reactions are known as leaching.
Organic chemicals like petroleum products also pollute many sources of water e.g., major oil spills in oceans.
(ii) Pesticides: These are mostly chlorinated hydrocarbons, organophosphates metallic salts etc. They dissolve in water to a small extent and pollute it. Since all the pesticides are toxic in nature, they are injurious to both plants and animals.
(iii) Polychlorinated biphenyls (PCBs): These are the chemical compounds used as fluids in transformers and capacitors. These are released into the atmosphere as vapours. They mix with rainwater and thus contaminate the water.
(iv) Eutrophication: The process in which algae-like organisms reduce dissolved oxygen in water is called eutrophication. It is harmful to aquatic life.
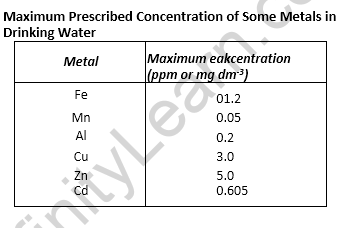
• International Standards for Drinking Water
Fluoride: The concentration of fluoride up to 1 ppm or 1 mg dm-3, is not harmful to human
beings if it is used as drinking water. The F~ ions make the enamel on teeth much harder by converting hydroxyapatite [3Ca3(P04)2- Ca(OH)2] the enamel on the surface of the teeth, into much harder fluorapatite, [3Ca3(P04)2- CaF2]. The concentration of F~ above 2 ppm causes brown mottling of teeth. Excess of fluoride is harmful to bones also.
Lead: Upper limit concentration of lead in drinking water is about 50 ppm. Lead can damage kidneys, liver, reproductive systems etc.
Sulphate: At a moderate level it is harmless but excess is harmful.
Nitrate: The maximum limit of nitrate should be 50 ppm. Excess nitrate in drinking water can cause diseases such as methemoglobinemia (blue baby syndrome).
Chemical Oxygen Demand (COD): Water is treated with K2Cr207 in an acidic medium to oxidise polluting substances which cannot be oxidised by microbial oxidation. The remaining is determined by back titration with a suitable reducing agent.
From the concentration of K2Cr207 consumed, the amount of O2 used in the oxidation is calculated.
![]()
• Soil Pollution—Sources of Soil Pollution
Pesticides: It can be classified as:
(i) Insecticide: The most common insecticides are chlorinated hydrocarbons like DDT, BHC etc.
As they are not much soluble in water, they stay in the soil for a long time. They are ‘ absorbed by the soil and contaminate root crops like radish, carrot etc.
(ii) Herbicides: These are the compounds used to control weeds, namely, sodium chlorate (NaCl03) and sodium arsenite (Na3As03) are commonly used herbicides but arsenic compounds, being toxic are no longer preferred.
Fungicides: Organo-mercury compounds are the most common fungicides. Its dissociation in soil produces mercury which is highly toxic and harmful for the crops. I Industrial Waste: It has been seen that most of the industrial wastes are thrown into water or dumped into the soil. These industrial wastes contain huge amounts of toxic chemicals which are mostly non-biodegradable. For example, metal processing industries, mining cement, glass industries, petroleum industry etc., fertilizer industry produce gypsum.
The disposal of non-biodegradable industrial solid waste is not done by suitable methods i and cause many serious problems.
Strategies to control environmental pollution:
(i) The improper disposal of wastes is one of the major causes of environmental degradation. The management of waste is very important.
(ii) All domestic wastes should be properly collected and disposed of.
• Green Chemistry
Green Chemistry is a way of thinking and is about utilising the knowledge and principles of
the chemistry that would control the increasing environmental pollution.
Green chemistry in day-to-day life:
(i) Dry-Cleaning of clothes and laundry: Replacement of halogenated solvent like (CCl4) by liquid C02 which is less harmful to groundwater.
Hydrogen peroxide (H202) is used for the purpose of bleaching clothes.
(ii) Bleaching of Paper: In place of chlorine H202 is used for the bleaching of paper,
(iii) Synthesis of Chemicals: Ethahal (CH3CHO) is prepared by step oxidation of ethene. Such as,
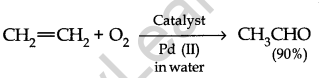
• Environmental pollution: It is the effect of undesirable changes in the surroundings that have harmful effects on plants, animals, and human beings.
• Troposphere: The lowest region of atmosphere which extends up to the height of 10 km from sea level in which man and other living organisms exist.
• Stratosphere: It is above the troposphere between 10 to 50 km above sea level.
• Acid rain: It is caused by the presence of oxides of sulphur nitrogen and C02 in the atmosphere. The pH of the raindrops is below 5.6, and it becomes acidic.
• Greenhouse gases: Some gases like carbon dioxide, methane, ozone, water vapours, CFCs have the capacity to trap some of the heat radiations from the earth or from the sun. This leads to global warming.
• Eutrophication: When phosphate ion increases in water it increases the growth of algae which consume the dissolved oxygen in water consequently aquatic life is adversely affected. This results in loss of biodiversity and the phenomenon are known as Eutrophication.
• COD (Chemical Oxygen Demand): It is calculated as the amount of oxygen required to oxidise the polluting substances. It is measured by treating the given sample of water with an oxidising agent, generally K2Cr207in the presence of oil. H2S04.

