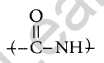Table of Contents
Time allowed: 3 hours
Maximum Marks: 70
CBSE Previous Year Question Papers Class 12 Chemistry 2012 Delhi Set I
Question 1.
What is meant by ‘doping’ in a semiconductor? [1]
Question 2.
What is the role of graphite in the electrometallurgy of aluminium? [1]
Answer:
Graphite is used as anode and useful for the reduction of Al2O3 into Al.
Question 3.
Which one of PCl–4 and PCl+4 is not likely to exist and why? [1]
Question 4.
Give the IUPAC name of the following compound.
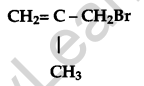
Answer:
3-Bromo-2-methylpropene
Question 5.
Draw the structural formula of a 2-methylpropan-2 ol molecule. [1]
Answer:
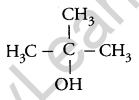
Question 6.
Arrange the following compounds in increasing order of their reactivity in nucleophilic addition reactions:
ethanol, propanal, propanone, butanone.
Answer:
Butanone < Propanone < Propanol < Ethanol
Question 7.
Arrange the following in the decreasing order of their basic strength in aqueous solutions: [1]
CH3NH2, (CH3)2NH, (CH3)3N and NH3
Answer:
(CH3)2NH > CH3NH2 > (CH3)3N > NH3
Question 8.
Define the term, ‘homopolymerization’ giving an example. [1]
Answer:
Polymerisation involving the presence of only one monomer is called homopolymerization.
e.g., polyethene is a homopolymer
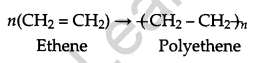
Question 9.
A 1.00 molal aqueous solution of trichloroacetic acid (CCl3COOH) is heated to its boiling point. The solution has a boiling point of 100.18 °C. Determine the Van’t Hoff factor for trichloroacetic acid. (Kb for water = 0.512 K kg mol-1) [2]
OR
Define the following terms:
(i) Mole fraction
(ii) Isotonic solutions
(iii) Van’t Hoff factor
(iv) Ideal solution
Answer:

(i) The ratio of the number of moles of a component in a mixture to the total number of moles in the mixture is called the mole fraction of that component. It is denoted by ‘x’.
(ii) Two solutions having the same molar concentration are said to be isotonic solutions. e.g., All intravenous injections must be isotonic with body fluids.
(iii) The ratio of observed colligative property to the calculated colligative property is called the Van’t Hoff factor. It is denoted by ‘i’.
(iv) Solutions that follow Raoult’s law at all temperatures and concentrations are called ideal solutions.
Question 10.
What do you understand by the ‘order of a reaction’? Identify the reaction order from each of the following units of reaction rate constant: [2]
- L-1 mol s-1
- L mol-1 s-1
Answer:
The sum of the powers to which the concentration of reactants are raised in the rate law expression is called the order of a reaction.
- Zero-order reaction
- Second-order reaction.
Question 11.
Name the two groups into which the phenomenon of catalysis can be divided. Give an example of each group with the chemical equation involved. [2]
Answer:
Catalysis can be positive, that is, it increases the rate of the reaction or negative i.e., decreases the rate of reaction. Depending on the phase of the reactants and the catalyst, catalysis can be:
(i) Homogenous Catalysis: The reactants and catalyst are in the same phase.
![]()
(ii) Heterogeneous catalysis: The reactants and catalyst are in a different phase.
![]()
Question 12.
What is meant by coagulation of a colloidal solution? Describe briefly any three methods by which coagulation of lyophobic sols can be carried out. [2]
Answer:
The process of setting of colloidal particles is called coagulation of sol. Methods of coagulation are:
(i) Electrophoresis: In this process, the colloidal particles move towards oppositely charged electrodes and get discharged and precipitated.
(ii) Mixing two Oppositely Charged Sols: Equal proportions of oppositely charged sols are mixed, they get neutralized and get precipitated.
(iii) Dialysis: Electrolytes are removed from the sol and colloid becomes unstable and gets coagulated.
Question 13.
Describe the principle involved in each of the following processes. [2]
(i) Mond process for refining of Nickel.
(ii) Column chromatography for purification of rare elements.
Answer:
(i) Nickel combined with carbon monoxide to form a volatile complex which is further decomposed to get back a pure nickel.
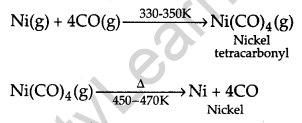
(ii) The basic principle involved in column chromatography is that different elements present in a mixture are adsorbed on adsorbent at different extents.
Question 14.
Explain the following giving an appropriate reason in each case. [2]
(i) O2 and F2 both stabilize higher oxidation states of metals but O2 exceeds F2 in doing so.
(ii) Structures of xenon fluorides cannot be explained by Valence Bond approach.
Answer:
(i) Due to the difference in the atomic size of oxygen and fluorine and the property of oxygen to form multiple bonds with metals, O2 exceeds F2, stabilize higher oxidation states.
(ii) For explaining the structures of xenon fluorides, we need to use VSEPR and hybridization theories because, in VBT, covalent bonds are formed by overlapping of a half-filled atomic orbital. But xenon has fully filled electronic configuration.
Question 15.
Complete the following chemical equations: [2]
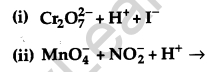
Answer:
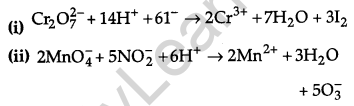
Question 16.
What is meant by
(i) peptide linkage
(ii) biocatalysts? [2]
Answer:
(i) Peptides linkage is the amide bond that helps to connect amino acids to form proteins. It is formed between -COOH and -NH2 group of two amino acids with the loss of water molecule.
(ii) Biocatalysts are enzymes that catalyse the biochemical reactions in the bodies of living organisms, e.g., Amylase.
Question 17.
Write any two reactions of glucose which cannot be explained by the open chain structure of the glucose molecule. [2]
Answer:
Two reactions which can’t be explained by the open chain structure of glucose are:
- Despite having the aldehyde group, glucose does not give 2, 4 -DNP test.
- The pentaacetate of glucose does not react with hydroxylamine indicating the absence of free -CHO group.
Question 18.
Draw the structure of the monomer for each of the following polymers: [2]
(i) Nylon 6
(ii) Polypropene.
Answer:
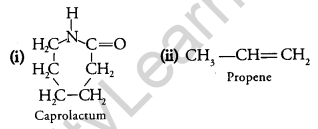
Question 19.
Tungsten crystallizes in a body-centred cubic unit cell. If the edge of the unit cell is 316.5 pm, what is the radius of the tungsten atom? [3]
OR
Iron has a body-centred cubic unit cell with a cell dimension of 286.65 pm. The density of iron is 7.874 g cm-3. Use this information to calculate Avogadro’s number. (Atomic mass of Fe = 55.845 u)
Question 20.
Calculate the amount of KCl which must be added to 1 kg of water so that the freezing point is depressed by 2K. (Kf for water = 1.86 K kg mol-1) [3]
Answer:

Question 21.
For the reaction [3]
2NO(g) + Cl2(g) → 2NOCl(g)
the following data were collected. All the measurements were taken at 263 K:
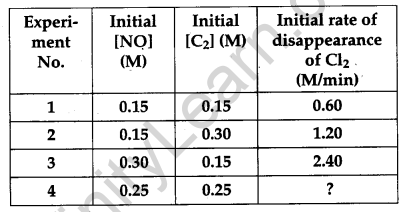
(a) Write the expression for rate law.
(b) Calculate the value of rate constant and specify its units
(c) What is the initial rate of disappearance of Cl2 in exp. 4?
Answer:
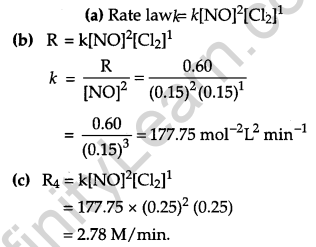
Question 22.
How would you account for the following? [3]
(i) Many of the transition elements are known to form interstitial compounds.
(ii) The metallic radii of the third (5d) series of transition metals are virtually the same as those of the corresponding group members of the second (4d) series.
(iii) Lanthanoids form primarily +3 ions, while the actinoids usually have higher oxidation states in their compounds, +4 or even +6 being typical.
Answer:
(i) Transition metal lattice has voids and hence these voids can trap small atoms like H, C and N to form interstitial compounds.
(ii) It is because electrons first fill the 4f orbitals and then the 5d-orbitals causing a decline in radii of third (5d) series.
(iii) This is because of the comparable energies of 5f, 6d and 7s orbitals so all of them can participate
Question 23.
Give the formula of each of the following coordinate with BaSO4: [3]
(i) Co3+ ion is bound to one Cl–, one NH3 molecule and two bidentate ethylenediamine (en) molecules.
(ii) Ni2+ ion is bound to two water molecules and two oxalate ions.
Write the name and magnetic behaviour of each of the above coordination entities.
(At. Nos. Co = 27, Ni = 28)
Answer:
(i) [Co(NH3)(en)2Cl]2+
Amminebis (ethylenediamine) chloride cobalt (III) ion, diamagnetic.
(ii) [Ni(H2O)2(C2O4)2]2-
Diaqua Oxalato Nickelate (II) ion, paramagnetic.
Question 24.
Although chlorine is an electron-withdrawing group, yet it is ortho, para directing in electrophilic aromatic substitution reactions. Explain why it is so? [3]
Answer:
Due to resonance, the electron density is increased on ortho and para positions for electron-donating (via resonance) groups like chlorine. Thus chlorine is an electron-withdrawing group yet it electrophilic is ortho, para-direction in an aromatic substitution reaction.
Question 25.
Draw the structure and name the product formed if the following alcohols are oxidized. Assume that an excess of an oxidizing agent is used. [3]
(i) CH3CH2CH2CH2OH
(ii) 2-butanol
(iii) 2-methyl-1-propanol
Answer:

Question 26.
Write chemical equations for the following conversions: [3]
(i) Nitrobenzene to benzoic acid.
(ii) Benzyl chloride to 2-phenylethanolamine.
(iii) Aniline to benzyl alcohol.
Answer:
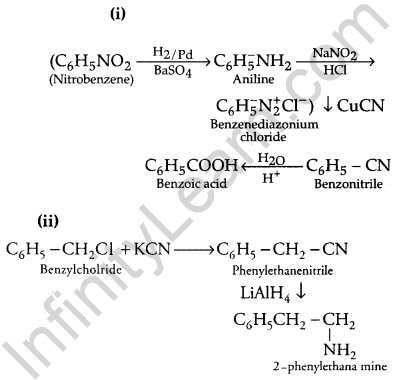
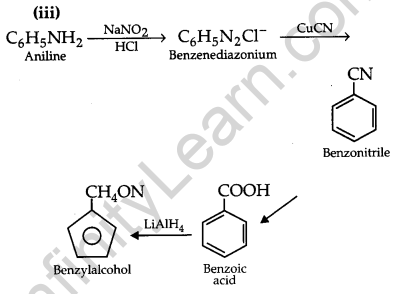
Question 27.
What are the following substances? Give one example of each one of them. [3]
(i) Tranquilisers
(ii) Food preservatives
(iii) Synthetic detergents.
Answer:
(i) Tranquilisers are drugs that act on the central nervous system to get relief from anxiety, stress etc. They are used in the treatment of stress-related mental disorders eg., Equanil.
(ii) These are chemicals used to preserve food by protecting it against microbial growth, e.g., Sodium benzoate.
(iii) Synthetic detergents have all the properties of soap but they do not precipitate in hard water. e.g., Sodium p-dodecyl benzene sulphonate.
Question 28.
(a) What type of battery is the lead storage battery? Write the anode and the cathode reactions and the overall reactions occurring in a lead storage battery when current is drawn from it. [2], [3]
(b) In the button cell, widely used in watches, the following reaction takes place.
Zn(s) + Ag2O(s) + H2O(l) → Zn2+(aq) + 2Ag(s) + 2OH–(aq)
Determine E0 and ΔG0 for the reaction (Given: E0(Ag+/Ag) = +0.80V, E0(Zn2+/Zn) = -0.76 V)
OR
(a) Define molar conductivity of a solution and explain how molar conductivity changes with change in concentration of solution for a weak and a strong electrolyte.
(b) The resistance of a conductivity cell containing 0.001 M KCl solution at 298 K is 1500 Ω. What is the cell constant if the conductivity of 0.001 M KCl solution at 298 K is 0.146 × 10-3 S cm-1?
Answer:
(a) The lead storage battery is secondary cell and can be recharged by passing current through it.

OR
(a) Molar conductivity of a solution is the conductance of the solution with one mole of electrolyte placed between electrodes 1 cm apart. With dilution, molar conductivity of weak electrolytes increases sharply and for strong electrolytes also, it increases.

Question 29.
(a) Complete the following chemical reactions, equations: [2], [3]
(i) P4 + SO2Cl2 →
(ii) XeF6 + H2O →
(b) Predict the shape and asked the angle (90° or more or less) in each of the following cases:
(i) SO2-3 and the angle O-S-O
(ii) ClF3 and the angle F-Cl-F
(iii) XeF2 and the angle F-Xe-F
OR
(a) Complete the following chemical equations:
(i) NaOH + Cl2 →
(hot and conc.)
(ii) XeF4 + O2F2 →
(b) Draw the structures of the following molecules:
(i) H3PO2
(ii) H2S2O7
(iii) XeOF4
Answer:
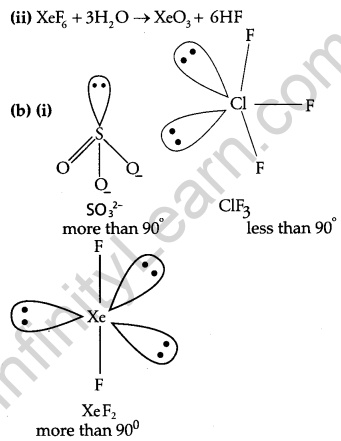
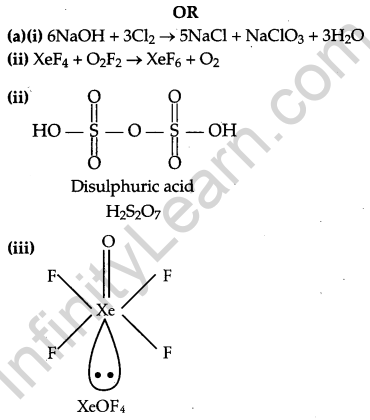
Question 30.
(a) Illustrate the following name reactions giving a suitable example in each case: [2],[3]
(i) Clemmensen reduction
(ii) Hell-Volhard-Zelinsky reaction
(b) How are the following conversions carried out?
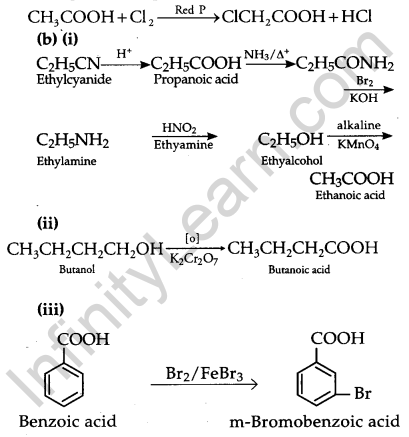
(i) Ethyl Cyanide to ethanoic acid
(ii) Butan-1-ol to butanoic acid
(iii) Benzoic acid to m-bromobenzoic acid
OR
(a) Illustrate the following reactions giving a suitable example for each:
(i) Cross aldol condensation
(ii) Decarboxylation
(b) Give simple tests to distinguish between the following pairs of compounds:
(i) Pentan-2-one and Pentan-3-one
(ii) Benzaldehyde and Acetophenone
(iii) Phenol and Benzoic acid
Answer:
(a) (i) Aldehydes and ketones are reduced to the CH2 group on treatment with zinc- amalgam and cone. HCl.
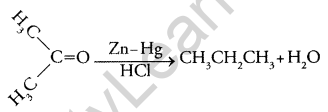
(ii) Carboxylic acids containing α-hydrogen atom gives halo carboxylic acids on treatment with halogens in the presence of red phosphorous.
OR
(a) (i) When aldol condensation is carried out between two different aldehydes or ketones it is called cross aldol condensation.
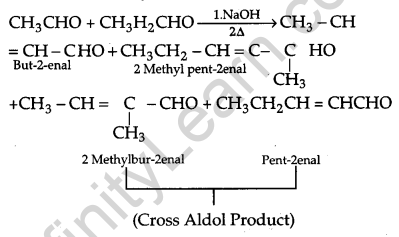
(ii) Carboxylic acid loses carbon dioxide to form hydrocarbon when their salts are heated with sodium. The reaction is known as decarboxylation reaction
![]()
(b) (i) Pentan-2-one gives iodoform test but pentan-3-one does not.
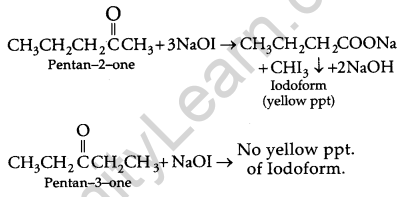
(ii) Benzaldehyde does not give iodoform test but Acetophenone gives iodoform test.

CBSE Previous Year Question Papers Class 12 Chemistry 2012 Delhi Set II
Note: Except for the following questions, all the remaining questions have been asked in the previous set.
Question 1.
Write a point of distinction between a metallic solid and an ionic solid other than metallic lustre. [1]
Question 11.
Describe a conspicuous change observed when: [2]
- a solution of NaCl is added to a sol of hydrated ferric oxide.
- a beam of light is passed through a solution of NaCl and then through a sol.
Answer:
- Coagulation of ferric hydroxide sol. would take place.
- NaCl solution is transparent so when a beam of light is passed, no Tyndall effect is produced. But on passing through the solution the path of light becomes visible due to the Tyndall effect.
Question 13.
Describe the following: [2]
(i) The role of cryolite in electrometallurgy of aluminium.
(ii) The role of carbon monoxide in the refining of crude nickel.
Answer:
(i) Cryolite lowers the melting point of the mixture and brings conductivity. Therefore, it is mixed with alumina during the metallurgy of aluminium.
(ii) Carbon monoxide forms a volatile complex with nickel. The volatile complex is then subjected to a high temperature to get pure metal through decomposition.
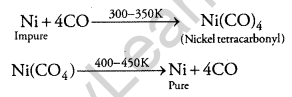
Question 14.
What is meant by
- peptide linkage
- biocatalysts? [2]
Answer:
- Peptide Linkage: It is the linkage between amino acids, formed due to loss of a water molecule and amide linkage is formed.

- Biocatalysts: Biocatalysts are enzymes which are used to perform chemical reactions on organic compounds.
Question 18.
Write the main structural difference between DNA and RNA. Of the two bases, thymine and uracil, which one is present in DNA? [2]
Answer:
| S.No. | DNA | RNA |
| 1. | DNA has deoxyribose sugar. | RNA has ribose sugar. |
| 2. | It has a double-helical structure. | It is single-stranded. |
| 3. | It is less reactive due to the presence of C-H bonds. | It has larger grooves so it is more reactive. |
DNA contains thymine base and uracil base is present in RNA.
Question 23.
How would you account for the following? [3]
(i) With the same d-orbital configuration (d4) Cr2+ is a reducing agent while Mn3+ is an oxidizing agent.
(ii) The actinides exhibit a larger number of oxidation states than the corresponding members in the lanthanide series.
(iii) Most of the transition metal ions exhibit characteristic in colours in aqueous solutions.
Answer:
(i) Cr2+ has d4 configuration. It gets oxidized to Cr3+ with electronic configuration d3 which is more stable. Therefore, Cr2+ is a reducing agent. Mn3+ has d4 configuration. It gets reduced to Mn2+ with d5 configuration. This is half-filled d-orbital and is stable. Therefore Mn3+ is an oxidizing agent.
(ii) Because 5f, 6d and 7s energy levels have a small energy gap in the actinide series. Due to these orbitals, actinoids exhibit a large number of oxidation states.
(iii) Due to partial absorption of visible light, the electron from one orbital gets promoted to another orbital of the d subshell. Due to the presence of impaired electrons transition metals are coloured.
Question 30.
(a) Give a possible explanation for each one of the following: [2], [3]
(i) There are two -NH2 groups in semicarbazide. However, only one such group is involved in the formation of semicarbazones.
(ii) Cyclohexanone forms cyanohydrin in good yield but 2, 4, 6-trimethylcyclohexanone does not.
(b) An organic compound with molecular formula C9H10O forms 2, 4, -DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro’s reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
OR
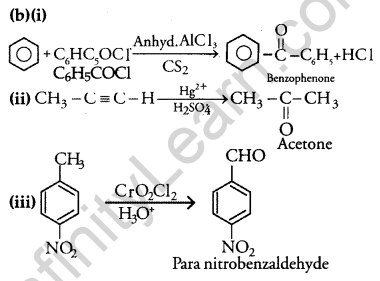
(a) Give chemical tests to distinguish between
(i) Phenol and Benzoic acid
(ii) Acetophenone and Benzophenone
(b) Write the structures of the main products of the following reactions:
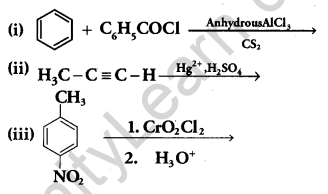
Answer:
(a) (i) Semicarbazide show resonance involving one of the two -NH2 groups, which is attached to the carboxyl carbon atom. Due to which electron density on -NH2 group involved in resonance decreases. So it cannot act as a nucleophile. Other -NH2 group can act as a nucleophile to produce semicarbazones with aldehydes and ketones.
(ii) In cyclohexanone CN- can easily attack without any steric hindrance. But in 2, 4, 6 – Trimethylcyclohexanone due to the presence of methyl groups steric hindrance is produced and CN- cannot attack effectively.
(iii) C9H10O is aldehyde because it reduces Tollen’s reagent. It undergoes Cannizaro’s reaction, therefore, it is substituted benzaldehyde. It gives 1, 2, Benzene-dicarboxylic acid. The compound is 2-Ethylbenzaldehyde.
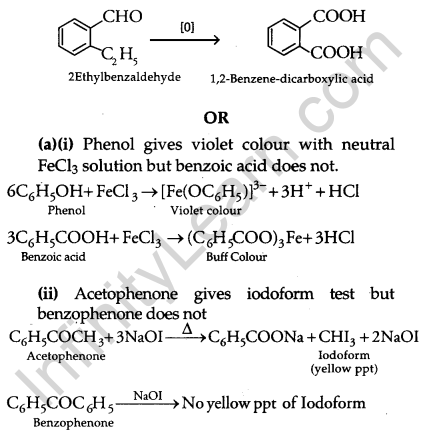
CBSE Previous Year Question Papers Class 12 Chemistry 2012 Delhi Set III
Note: Except for the following questions, all the remaining questions have been asked in previous sets.
Question 3.
Out of PH3 and H2S, which is more acidic and why? [1]
Answer:
H2S is more acidic than PH3 due to a smaller size and higher electronegativity of sulphur. Therefore S-H bond is polar than P-H bond and easy to remove.
Question 5.
Draw the structure of a hex-1-en-3-ol compound. [1]
Answer:
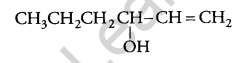
Question 12.
Explain the following terms giving one example for each: [2]
(i) Micelles
(ii) Aerosol.
Answer:
(i) Micelles are aggregates which exhibit colloidal behaviour at higher concentration, with the hydrophilic part outside and the hydrophobic part towards the oil and dirt particle, e.g., soap
(ii) An aerosol is a colloid of the fine solid particle or liquid drops in the air or another gas. It can be natural or artificial, e.g. dust particle and smoke.
Question 20.
15.0 g of an unknown molecular material was dissolved in 450 g of water. The resulting solution was found to freeze at -0.34 °C. What is the molar mass of this material? (Kf for water = 1.86 K kg mol-1) [3]
Answer:

Question 22.
Explain the following observations giving an appropriate reason for each: [3]
- The enthalpies of atomization of transition elements are quite high.
- There occurs much more frequent metal-metal bonding in compounds of heavy transition metals (i.e., 3rd series).
- Mn2+ is much more resistant than Fe2+ towards oxidation.
Answer:
- Due to the presence of metallic bonds as a result of a large number of valence electrons.
- The presence of valence electrons and impaired d-orbital electrons help heavy transition metals to form metallic bonds.
- Due to the stability of Mn2+ because of half filled-subshell (3d5), it does not gets oxidized. But Fe2+ has 3d6 configuration and it can lose one electron to become 3d5 which is stable. Therefore it is easily oxidized.
Question 23.
Write the name, the structure and the magnetic behaviour of each of the following complexes: [3]
- (Pt(NH3)Cl(NO2)]
- (CO(NH3)4Cl2]Cl
- Ni(CO)4
(At. Nos. Co = 27, Ni = 28, Pt = 78)
Answer:
- Amminechloridonitrito-N-platinum
- trigonal planar, diamagnetic (II) Tetra-ammine dichloride cobalt (III) chloride, octahedral, diamagnetic
- Tetracarbonyl nickel (0), Tetrahedral, diamagnetic
Question 27.
Explain the following terms giving one example of each type: [3]
- Antacids,
- Disinfectants,
- Enzymes.
Answer:
- Substances consumed to reduce acidity in the stomach by neutralizing excess HCl produced by the stomach, e.g. Milk of magnesia.
- Disinfectants are chemicals used to kill microorganism, applied only to non-living objects like floors and drains, e.g. 1% phenol solution.
- Enzymes are structurally globular proteins that catalyse biochemical reactions in living organisms e.g. Trypsin.
Question 30.
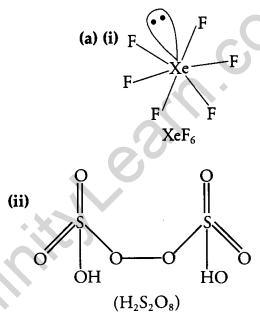
Draw the molecular structures of the following compounds: [2], [3]
(i) XeF6
(ii) H2S2O8
(b) Explain the following observations:
(i) The molecules NH3 and NF3 have dipole moments which are of the opposite direction.
(ii) All the bonds of PCl5 molecule are not equivalent.
(iii) Sulphur in vapour state exhibits paramagnetism.
OR
(a) Complete the following chemical equations:
(i) XeF4 + SbF5 →
(ii) Cl2 + F2 (excess) →
(b) Explain each of the following:
(i) Nitrogen is much less reactive than phosphorus.
(ii) The stability of the +5 oxidation state decreases down group 15.
(iii) The bond angles (O-N-O) are not of the same value in NO–2 and NO+2.
Answer:
(b) (i) Fluorine is more electronegative than nitrogen while hydrogen is less electronegative than nitrogen resulting in opposite dipole moments of NH3 and NF3. Dipole points towards N in NH3 and towards F in NF3.
OR
(a) (i) XeF4 + SbF5 → [XeF3]+[SbF6]–
(ii) Cl2 + 3F2 (excess) → 2ClF3