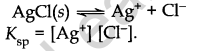Chapter 7 of the Class 11 Chemistry notes focuses on the fundamental concept of equilibrium. In this chapter, you will delve into the principles governing chemical equilibrium, which is a state where the forward and reverse reactions occur at equal rates, resulting in a constant composition over time.
The notes cover the essential aspects of equilibrium, including the equilibrium constant, its mathematical representation, and its applications. You will also explore Le Chatelier’s principle, which explains how a system at equilibrium responds to changes in concentration, pressure, or temperature.
By mastering the concepts presented in these notes, you will develop a strong foundation in understanding the dynamic nature of chemical reactions and their equilibrium states. This knowledge will be crucial for your success in advanced chemistry topics and problem-solving.
- Chemical Equilibrium
In a chemical reaction chemical equilibrium is defined as the state at which there is no further change in concentration of reactants and products.
For example,
![]()
At equilibrium, the rate of the forward reaction is equal to the rate of the backward reaction. Equilibrium mixture: The mixture of reactants and products in the equilibrium state is called an equilibrium mixture.
Based on the extent to which the reactions proceed to reach the state of equilibrium, these may be classified into three groups:
(i) The reactions proceed almost to completion and the concentrations of the reactants left are negligible.
(ii) The reactions in which most of the reactants remains unchanged, i.e. only small amounts of products are formed.
(iii) The reactions in which the concentrations of both the reactants and products are comparable when the system is in equilibrium.
• Equilibrium in Physical Processes
(i) Solid-Liquid Equilibrium: The equilibrium is represented as
![]()
Rate of melting of ice = Rate of freezing of water.
The system here is in dynamic equilibriums and the following can be inferred.
(a) Both the opposing processes occur simultaneously
(b) Both the processes occur at the same rate so that the amount of ice and water – remains constant.
(ii) Liquid-Vapour Equilibrium
The equilibrium can be represented as
![]()
Rate of evaporation = Rate of condensation
When there is an equilibrium between liquid and vapours, it is called liquid-vapour equilibrium.
- Solid-Vapour Equilibrium
This type of equilibrium is attained where solids sublime to the vapour phase. For example, when solid iodine is placed in a closed vessel, violet vapours start appearing in the vessel whose intensity increases with time and ultimately, it becomes constant.

- Equilibrium involving Dissolution of Solid in Liquid
Solution: When a limited amount of salt or sugar or any solute dissolves in a given amount of water solution is formed.
At a given temperature state is reached when no more solute can be dissolved then the solution is called a saturated solution.
The equilibrium between a solid and its solution is indicated by the saturated solution and may be represented as
![]()
Here dissolution and precipitation take place at the same speed.
On adding a small amount of radioactive sugar to the saturated solution it will be found that the sugar present in the solution as well as in the solid-state is radioactive.\
- Equilibrium between a Gas and its Solution in Liquid
This type of equilibrium can be seen by the following example:
Let us consider a sealed soda water bottle in which C02 gas is dissolved under high pressure. A state of equilibrium is attained between CO2 present in the solution and the vapours of the gas.
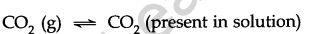 Henry’s law: The solubility of a gas in a liquid at a certain temperature is governed by Henry’s law. It states that the mass of a gas that dissolves in a given mass of a solvent at any temperature is proportional to the pressure of the gas above the surface of the solvent.
Henry’s law: The solubility of a gas in a liquid at a certain temperature is governed by Henry’s law. It states that the mass of a gas that dissolves in a given mass of a solvent at any temperature is proportional to the pressure of the gas above the surface of the solvent.
![]()
• Characteristics of Equilibria Involving Physical Processes
(i) The equilibrium can be attained only in closed systems at a given temperature.
(ii) At the equilibrium the measurable properties of the system remain constant.
(iii) The equilibrium is dynamic since both the forward and backward processes occur at the same rate.
(iv) At equilibrium, the concentrations of substances become constant at a constant temperature.
(v) The value of the equilibrium constant represents the extent to which the process proceeds before equilibrium is achieved.
- Equilibrium in Chemical Processes
Like equilibria in physical systems, it can also be achieved in a chemical process involving reversible chemical reactions carried in a closed container.
![]()
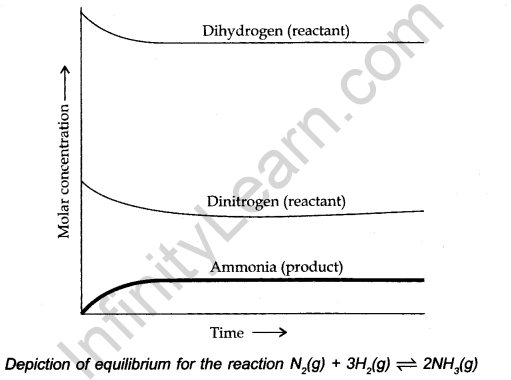
The dynamic nature of chemical equilibrium can be demonstrated in the synthesis of ammonia by Haber’s process. Haber started his experiment with the known amounts of N2 and H2 at high temperatures and pressure. At regular intervals of time, he determined the amount of ammonia present. He also found out the concentration of unreacted N2 and H2.
After a certain time, he found that the composition of the mixture remains the same even though some of the reactants are still present. This constancy indicates the attainment of equilibrium. In general, for a reversible reaction, the chemical equilibria can be shown by
![]()
After a certain time, the two reactions occur at the same rate and the system reaches a state of equilibrium. This can be shown by the given figure.
 Equilibrium in Homogeneous System
Equilibrium in Homogeneous System
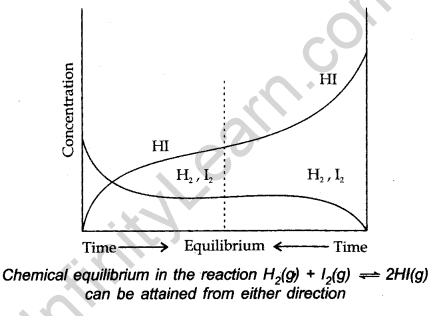
When in a system involving reversible reaction, reactants and products are in the same phase, then the system is called a homogeneous system.
For Example,
![]()
After some time it can be observed that equilibrium is formed. The equilibrium can be seen by constancy in the colour of the reaction mixture.
 Law of Chemical Equilibrium
Law of Chemical Equilibrium
At a constant temperature, the rate of a chemical reaction is directly proportional to the product of the molar concentrations of the reactants each raised to a power equal to the corresponding stoichiometric coefficients as represented by the balanced chemical equation. Let us consider the reaction,

• Relationship between Equilibrium constant K, reaction Quotient Q and Gibbs energy G.
A mathematical expression of the thermodynamic view of equilibrium can be described by the tine equation.
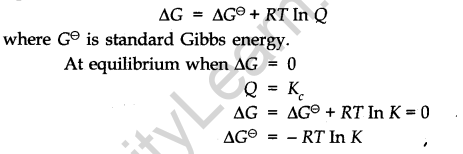
 Factors Affecting Equilibria
Factors Affecting Equilibria
- Le Chatelier’s principle: If a system under equilibrium is subjected to a change in temperature, pressure or concentration, then the equilibrium shifts in such a manner as to reduce or to counteract the effect of change.
- Effect of Change of Concentration: When the concentration of any of the reactants or products in a reaction at equilibrium is changed, the composition of the equilibrium changes to minimise the effect.
- Effect of Pressure Change
- If the number of moles of gaseous reactants and products is equal, there is no effect of pressure.
- When the total number of moles of gaseous reactants and a total number of moles of gaseous products are different.
- On increasing pressure, the total number of moles per unit volume increases, thus the equilibrium will shift in direction in which several moles per unit volume will be less.
- If the total number of moles of products is more than the total number of moles of reactants, low pressure will favour forward reaction.
- If the total number of moles of reactants is more than the total number of moles of products, high pressure is favourable to forward reaction.
- Effect of Inert Gas Addition
If the volume is kept constant there is no effect on equilibrium after the addition of inert gas.
Reason: This is because the addition of inert gas at constant volume does not change the partial pressure or the molar concentration.
The reaction quotient changes only if the added gas is involved in the reaction.
- Effect of Temperature Change
When the temperature of the system is changed (increased or decreased), the equilibrium shifts in the opposite direction to neutralize the effect of change. An exothermic reaction, low temperature favours forward reaction e.g.,
![]()
but practically very low temperature slows down the reaction and thus a catalyst is used. In the case of an endothermic reaction, the increase in temperature will shift the equilibrium in the direction of the endothermic reaction.
- Effect of a Catalyst
Catalyst does not affect the equilibrium composition of a reaction mixture.
Reason: Since catalyst increases the speed of both the forward and backward reactions to the same extent in a reversible reaction.
Ionic Equilibrium in Solution
- Electrolytes: Substances that conduct electricity in their aqueous solution.
- Strong Electrolytes: Those electrolytes which on dissolution in water are ionized almost completely are called strong electrolytes.
- Weak electrolyte: Those electrolytes which on dissolution in water partially dissociated are called a weak electrolytes.
- Ionic Equilibrium: The equilibrium formed between ions and unionised substance is called ionic equilibrium, e.g.,
![]()
Acids: Acids are the substances that turn blue litmus paper to red and liberate dihydrogen by reacting with some metals.
Bases: Bases are the substances that turn red litmus paper blue. It is bitter. Common Example: NaOH, Na2C03.
• Arrhenius Concept of Acids and Bases
Acids: According to Arrhenius theory, acids are substances that dissociate in water to give hydrogen ions H+(aq).
Bases: Bases are substances that produce OH–(aq) after dissociation in water.
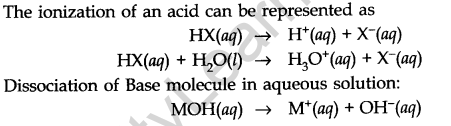 Limitations of the Arrhenius Concept
Limitations of the Arrhenius Concept
(i) According to the Arrhenius concept, an acid gives H+ ions in water but the H+ ions do not exist independently because of its very small size (~H-18 m radius) and intense electric field.
(ii) It does not account for the basicity of substances like ammonia which does not possess a hydroxyl group.
- The Bronsted-Lowry Acids and Bases
According to Bronsted-Lowry, an acid is a substance that is capable of donating a hydrogen ion H+ and bases are substances capable of accepting a hydrogen ion H+.
In other words, acids are proton donors and bases are proton acceptors. This can be explained by the following example.
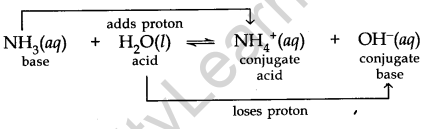 Acid and Base as Conjugate Pairs
Acid and Base as Conjugate Pairs
The acid-base pair that differs only by one proton is called a conjugate acid-base pair.
Let us consider the example of the ionization of HCl in water.
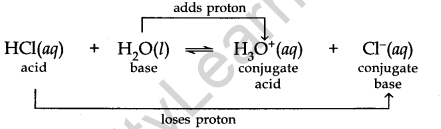
Here water acts as a base because it accepts the proton.
CL is a conjugate base of HCl and HCl is the conjugate acid of base CL. Similarly, H20 is the conjugate base of an acid H30+ and H30+ is the conjugate acid of base H2O.
- Lewis Acids and Bases
According to Lewis, acid is a substance that accepts an electron pair and base is a substance with donates an electron pair.
Electron deficient species like AlCl3, BH3, H+ etc. can act as Lewis acids while species like H20, NH3 etc. can donate a pair of electrons, can act as Lewis bases.
- Ionization of Acids and Bases
The strength of acid or base is determined with the help of the extent of ionization in an aqueous solution.
- pH Scale: Hydrogen-ion concentration is measured as the number of gram ions of hydrogen ions present per litre of solution. Since these concentrations are usually small, the concentration is generally expressed as the pH of the solution. pH is
the logarithm of the reciprocal of the hydrogen ion concentration.
 Di and Polybasic Acids
Di and Polybasic Acids
Acids that contain more than one ionizable proton per molecule are called Dibasic acids or polybasic acids or polyprotic acids. Common examples are oxalic acid, sulphuric acid, phosphoric acid etc.
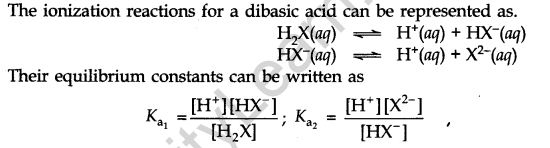
 Factors Affecting Acid Strength
Factors Affecting Acid Strength
When the strength of the H-A bond decreases
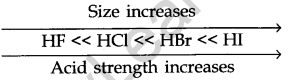
The energy required to break the bond decreases, H-A becomes a stronger acid.
As the size of A increases down the group, H-A bond strength decreases and so the acid strength increases.
In a period, as the electronegativity of A increases, the strength of the acid increases.
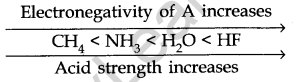
- Common Ion Effect
If in an aqueous solution of a weak electrolyte, a strong electrolyte is added having an ion common with the weak electrolyte, then the dissociation of the weak electrolyte is decreased or suppressed. The effect by which the dissociation of weak electrolytes is suppressed is known as the common ion effect.
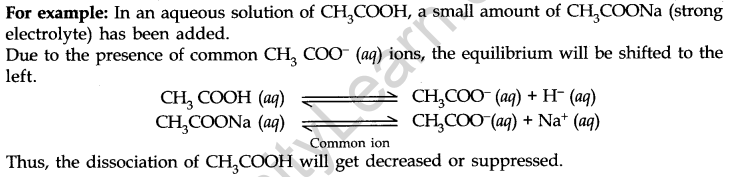 Hydrolysis of Salts and the pH of their Solutions
Hydrolysis of Salts and the pH of their Solutions
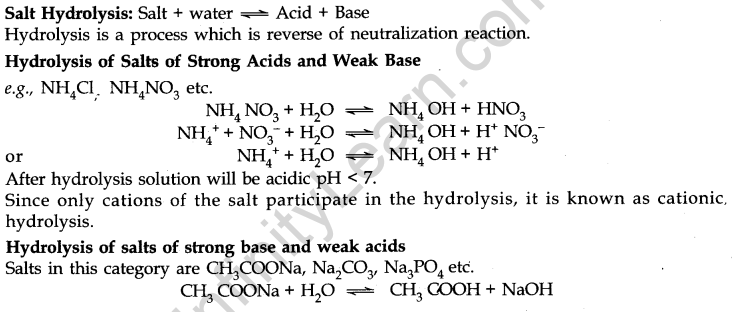

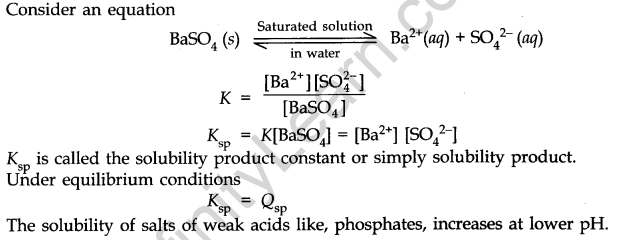
- Solubility Products
It applies to a sparingly soluble salt. There is an equilibrium between ions and unionised solid substances.
 Equilibrium: It can be established for both physical and chemical processes. At the state of the equilibrium rate of forwarding and backward reactions are equal.
Equilibrium: It can be established for both physical and chemical processes. At the state of the equilibrium rate of forwarding and backward reactions are equal.- Equilibrium constant: Kc is expressed as the concentration of products divided by reactants each term raised to the stoichiometric coefficients. For reactions,
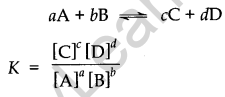 Le Chatelier’s principle: It states that the change in any factor such as temperature, pressure, concentration etc., will cause the equilibrium to shift in such a direction to reduce the effect of the change.
Le Chatelier’s principle: It states that the change in any factor such as temperature, pressure, concentration etc., will cause the equilibrium to shift in such a direction to reduce the effect of the change.- Electrolytes: Substances that conduct electricity in aqueous solutions are called electrolytes.
- Arrhenius Concept: According to Arrhenius, acids give hydrogenous while bases produce hydroxyl ions in their aqueous solution.
- Bronsted-Lowry concept: Bronsted-Lowry defined an acid as a proton donor and a base as a proton acceptor.
- Conjugate base and Conjugate acid: When a Bronsted-Lowry acid reacts with a base it produces its conjugate base and conjugate acid.\
- Conjugate pair of acid and base: Conjugate pair of acid and base differs only by one proton.
- Lewis acids: Define acid as an electron pair acceptor and a base as an electron-pair donor.
- pH Scale: Hydronium ion concentration in molarity is more conveniently expressed on a logarithmic scale known as the pH scale. The pH of pure water is 7.
- Buffer solution: It is the solution whose pH does not change by the addition of a small amount of strong acid or base.
For example CH3COOH + CH3COONa
- Solubility product (Ksp): For a sparingly soluble salt, it is defined as the product of the molar concentration of the ions raised to the power equal to the number of times each ion occurs in the equation for solubilities.