Table of Contents
Introduction Aluminium Hydroxide Formula
Aluminium hydroxide has the chemical formula Al(OH)3. It is an inorganic compound consisting of one aluminum atom bonded to three hydroxide ions.
Aluminium hydroxide is commonly found as a white, gelatinous solid. It is insoluble in water but can dissolve in acids and bases. It is widely used in various applications, including as an antacid to neutralize stomach acid, as an ingredient in certain medications, and as a flame retardant in plastics and coatings. Aluminium hydroxide is also used in water treatment processes to remove impurities and as a filler in the production of ceramics, paper, and rubber.
Structural Formula of Aluminium Hydroxide Formula
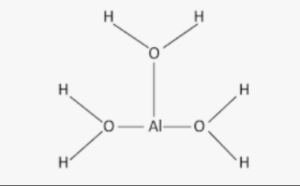
The structural formula of aluminium hydroxide is represented as Al(OH)3. It consists of an aluminum (Al) atom at the center, bonded to three hydroxide (OH) groups.
The hydroxide groups are composed of one oxygen (O) atom bonded to a hydrogen (H) atom. The structural formula shows the arrangement and bonding of atoms in the compound, providing a visual representation of its molecular structure.
Uses of Aluminium Hydroxide
- Antacid: Aluminium hydroxide is widely used as an antacid to neutralize excess stomach acid. It helps to relieve symptoms of heartburn, indigestion, and acid reflux by reducing the acidity in the stomach.
- Pharmaceuticals: Aluminium hydroxide is used as an active ingredient in certain medications, including over-the-counter and prescription drugs. It is used to treat conditions such as peptic ulcers and gastroesophageal reflux disease (GERD).
- Water Treatment: Aluminium hydroxide is used in water treatment processes to remove impurities and suspended particles. It acts as a flocculant and coagulant, helping to clarify water and improve its quality.
- Flame Retardant: Aluminium hydroxide is used as a flame retardant in various materials, including plastics, textiles, and coatings. It releases water vapor when exposed to fire, which helps to cool and suppress the flame.
- Industrial Applications: Aluminium hydroxide is used in various industrial processes, such as manufacturing ceramics, glass, and aluminum chemicals. It is also used as a filler in the production of rubber, plastics, and paper.
- Cosmetics: Aluminium hydroxide is used in cosmetic products, particularly in skincare formulations. It acts as a thickening agent, absorbent, and pH adjuster in products like lotions, creams, and foundations.
- Veterinary Medicine: Aluminium hydroxide is used in veterinary medicine as an adjuvant in vaccines. It helps to enhance the immune response and improve the effectiveness of the vaccine.
Physical Properties of Aluminium Hydroxide Formula
- Appearance: Aluminium hydroxide is a white, amorphous powder or solid.
- Density: The density of aluminium hydroxide is approximately 2.42 g/cm³.
- Melting Point: Aluminium hydroxide does not have a specific melting point, but it decomposes around 300°C (572°F).
- Solubility: Aluminium hydroxide is insoluble in water and organic solvents.
- pH: In aqueous solutions, aluminium hydroxide acts as a weak base and can increase the pH.
Chemical Properties of Aluminium Hydroxide Formula
- Acid-Base Reaction: Aluminium hydroxide acts as a base and can react with acids to form salts. It can neutralize strong acids, such as hydrochloric acid (HCl), to produce aluminium chloride (AlCl3) and water.
- Amphoteric Nature: Aluminium hydroxide exhibits amphoteric properties, meaning it can react both as a base and as an acid. It can react with strong bases, such as sodium hydroxide (NaOH), to form sodium aluminate (NaAlO2) and water.
- Dehydration: Aluminium hydroxide can undergo dehydration when heated, resulting in the formation of aluminium oxide (Al2O3) and water vapor. This process is often used to produce alumina, which is an important industrial material.
- Adsorption: Aluminium hydroxide has adsorption properties and can adsorb certain substances, including organic compounds, heavy metals, and toxins. This property makes it useful in wastewater treatment and purification processes.
- Reactivity with Other Compounds: Aluminium hydroxide can react with other compounds containing ions such as phosphate, sulfate, and carbonate, forming insoluble precipitates. These reactions are important in various industrial and environmental processes.
Conclusion
In conclusion, aluminium hydroxide (Al(OH)3) is a compound with diverse applications and benefits. Its chemical formula reflects its composition, consisting of aluminium ions (Al3+) and hydroxide ions (OH-) held together by ionic bonds. Aluminium hydroxide is widely used as an antacid to relieve symptoms of heartburn and indigestion. It acts by neutralizing excess stomach acid and providing relief to the digestive system. Additionally, aluminium hydroxide is employed as a phosphate binder in patients with kidney disease to reduce the levels of phosphates in the blood. It also finds application as a vaccine adjuvant to enhance immune responses and as a fire retardant in various materials to delay the spread of flames. Furthermore, aluminium hydroxide serves as a filler and pigment in industries such as plastics, paints, and coatings, improving strength, stability, and coloration. Its properties make it a valuable compound in various sectors, showcasing its versatility and significance in different applications.
Solved Examples on Aluminium Hydroxide Formula
Example 1: What is the balanced chemical equation for the reaction between aluminium hydroxide and hydrochloric acid?
Solution: The balanced chemical equation for this reaction can be written as:
2Al(OH)3 + 6HCl → 2AlCl3 + 6H2O
Example 2: When 50 grams of aluminium hydroxide reacts with excess sulfuric acid, how many grams of aluminium sulfate will be formed?
Solution: First, we need to calculate the molar mass of aluminium hydroxide:
Al(OH)3 = (1 × 26.98 g/mol) + (3 × 16.00 g/mol) + (3 × 1.01 g/mol) = 78.00 g/mol
Next, we calculate the number of moles of aluminium hydroxide:
moles = mass / molar mass = 50 g / 78.00 g/mol = 0.641 moles
From the balanced chemical equation, we can see that the molar ratio between aluminium hydroxide and aluminium sulfate is 1:1. Therefore, the number of moles of aluminium sulfate formed will also be 0.641 moles.
Finally, we calculate the mass of aluminium sulfate:
mass = moles × molar mass
= 0.641 moles × (1 × 26.98 g/mol + 1 × 32.07 g/mol + 4 × 16.00 g/mol)
= 54.7 g
So, 50 grams of aluminium hydroxide will produce 54.7 grams of aluminium sulfate.
Frequently Asked Questions on Aluminium Hydroxide Formula
What is aluminum hydroxide used for?
Aluminum hydroxide is used as an antacid to relieve heartburn, upset stomach, and indigestion.
Is Al OH 3 a base or acid?
Al OH 3 is a base because it can neutralize acids.
Is Al OH 3 a strong or weak base?
Aluminum hydroxide (Al OH 3) is a weak base.
Why is Al OH 3 called aluminum hydroxide?
Al OH 3 is called aluminum hydroxide because it contains aluminum (Al) and hydroxide (OH) ions in its chemical structure.
What is the main side effect of Aluminium containing antacids?
The main side effect of aluminum-containing antacids is constipation.
What happens if you take too much aluminum hydroxide?
Taking too much aluminum hydroxide can lead to aluminum toxicity, which may cause symptoms like confusion and bone problems.
What are the amphoteric properties of aluminium hydroxide?
Aluminum hydroxide is amphoteric, which means it can act as both an acid and a base depending on the conditions.
What are the properties of aluminium oxide?
Aluminum oxide is a white powder that is hard and has high thermal conductivity. It is commonly used as an abrasive and in various industrial applications.
What are the physical and chemical properties of aluminum hydroxide?
Aluminum hydroxide is a white, crystalline powder. It is insoluble in water but can react with acids. It is also amphoteric, meaning it can behave as both an acid and a base.








